BackgroundTirzepatide is an incretin-based medicine that activates both the glucagon-like peptide-1 and glucose-dependent insulinotropic peptide receptor. Its dual action has demonstrated reductions in blood glucose and body weight, but cardiovascular effects remain debated. In the SURPASS-CVOT trial, tirzepatide demonstrated non-inferiority to dulaglutide in reducing the risk of major adverse cardiovascular events, leaving uncertainty about its full cardioprotective effects. In this study, we evaluated the magnitude of tirzepatide’s effect on major adverse cardiovascular events compared with a validated placebo proxy in clinical practice.
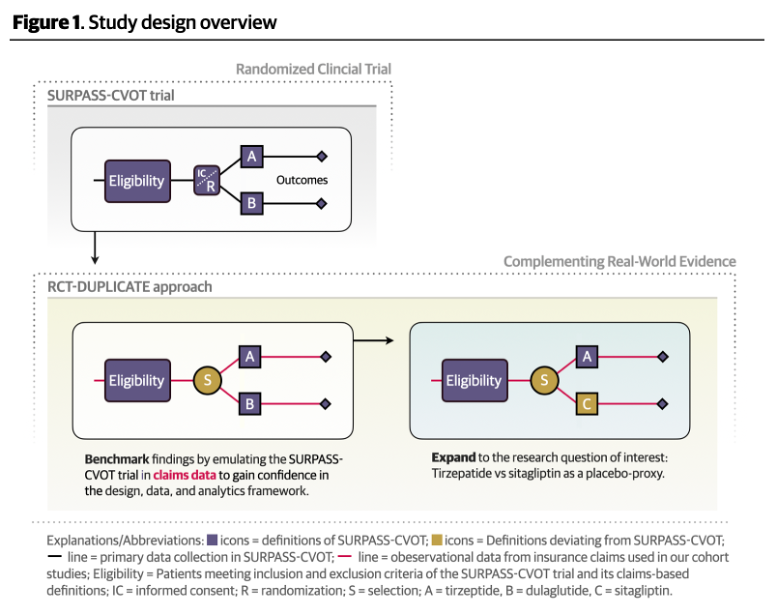
MethodsBuilding on insights from benchmarking against reference trials using the RCT-DUPLICATE approach, we conducted an active-comparator, new-user cohort study in patients aged 40 years or older with type 2 diabetes and atherosclerotic cardiovascular disease, emulating the eligibility criteria of SURPASS-CVOT. Tirzepatide initiators were compared with sitagliptin initiators, chosen as a placebo proxy based on prior evidence of neutral effects on cardiovascular end points. Propensity scores were estimated using comprehensive pre-treatment covariates with overlap weighting to achieve covariate balance. End points included major adverse cardiovascular events, and its individual components of myocardial infarction, stroke, or all-cause mortality. Hazard ratios (HRs) were estimated with weighted Cox models and pooled across databases using fixed-effects meta-analysis.
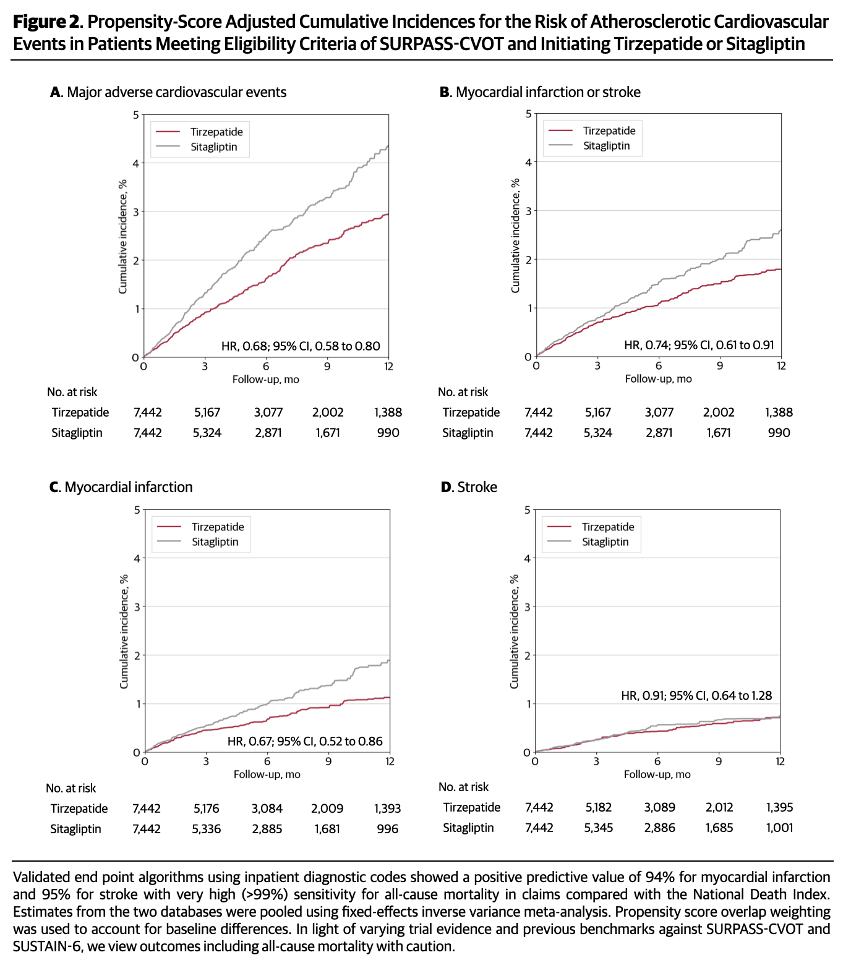
ResultsAfter weighting, baseline characteristics were well balanced between groups. Tirzepatide showed a lower risk of major adverse cardiovascular events (HR, 0.68; 95% CI, 0.58–0.80; NNT, 70) compared with sitagliptin, driven by a reduced risk of myocardial infarction (HR, 0.67; 95% CI, 0.52–0.87; NNT, 130) and all-cause mortality (HR, 0.55; 95% CI, 0.42–0.72; NNT, 122). In safety outcomes, serious bacterial infections were markedly reduced (HR, 0.64; 95% CI, 0.55–0.75; NNT, 48). The effectiveness of confounding control was supported by null effects observed for negative control outcomes. Sensitivity analyses yielded consistent results.
ConclusionsThese findings complement SURPASS-CVOT. Our approach provides a direct comparison of tirzepatide against a placebo proxy, providing clinicians and patients with estimates of how much tirzepatide reduces major adverse cardiovascular outcomes beyond standard background therapy in clinical practice. While awaiting accrual of further trial evidence for tirzepatide in cardiovascular indications, this study demonstrates how RCT-anchored real-world evidence can fill gaps for timely decision-making.
ClinicalTrials.gov Identifier: (NCT07203677)