Background. In acute heart failure (AHF), backward failure elevates systemic venous pressure, causing liver congestion and end-organ dysfunction. Clarifying the prognostic significance of this liver involvement is essential to optimize management and outcomes of AHF patients and associated liver dysfunction. We therefore investigated the prognostic impact of liver dysfunction in patients hospitalized with AHF, integrating echocardiographic and laboratory data.
Methods. We analyzed 370 adults hospitalized with AHF in an ongoing, prospective, single-center cohort initiated in 2019. Liver dysfunction was defined at hospitalization as ASAT ≥ 50 U/L or ALAT ≥ 50 U/L. Right heart failure was diagnosed based on clinical criteria, elevated natriuretic peptides (NT-proBNP ≥ 300 ng/l), and echocardiographic evidence of right ventricular dysfunction. Cox regression models were used to assess associations with all-cause mortality; stepwise models included additional adjustment for cardiac biomarkers. Kaplan-Meier curves were generated for all-cause mortality after hospitalization. Groups were compared using log-rank test. Univariable and multivariable (adjusted for age and sex) logistic regression analyses were fitted in order to investigate association between liver dysfunction and in-hospital cardiac worsening (defined as new/increased intravenous diuretics or inotropes, MCS, invasive ventilation, initiation of hemodialysis).
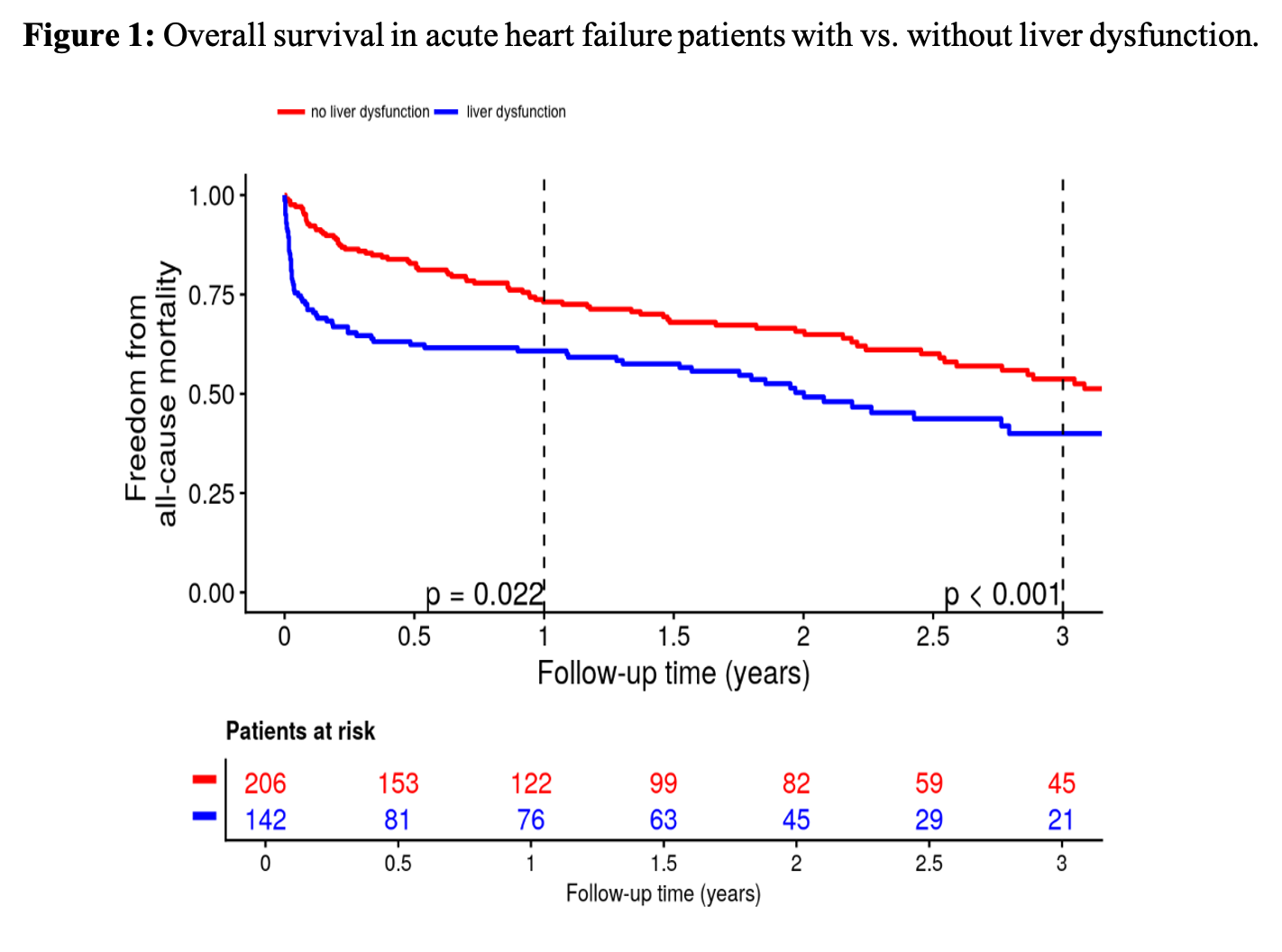
Results. Liver dysfunction was present in 41% of the 370 patients (median age 73 years; 69.5% men). Evidence of right heart failure was observed in 97%, consistent with biventricular involvement in acute decompensation. Liver dysfunction was associated with lower systolic and diastolic blood pressure (both p<0.001), impaired left ventricular function at baseline (p<0.001), and higher pro-adrenomedullin (p<0.001). Baseline TAPSE was reduced in both groups, with significantly lower values in the liver-dysfunction group (p<0.0064). By discharge, TAPSE no longer differed, paralleling a marked decline in transaminase levels in the liver-dysfunction group. Patients with liver dysfunction had higher 1-year and 3-year all-cause mortality (see Figure 1). In multivariable analyses, liver dysfunction predicted mortality in unadjusted analyses (HR 1.71, 95% CI [1.25, 2.33]; p<0.001) and after adjustment for age and sex (HR 1.82, 95% CI [1.32, 2.50]; p<0.001). The association remained significant after further adjustment for NT-proBNP (HR 1.98, 95% CI [1.34, 2.94]; p<0.001) and pro-adrenomedullin (HR 1.61, 95% CI [1.07, 2.40]; p=0.021), but not after adjustment for high-sensitivity troponin I (HR 1.20, 95% CI [0.75, 1.91]; p=0.45). Liver dysfunction was not associated with in-hospital cardiac worsening (unadjusted OR 1.46, 95% CI [0.91, 2.38], p=0.12; age-sex adjusted OR 1.39, 95% CI [0.86, 2.27], p=0.19).
Conclusions. Liver dysfunction is common in AHF patients and reversible upon decongestion. It is associated with increased risk of death and may therefore help to identify high-risk patients with AHF. Further research is needed to clarify mechanistic pathways and to define optimal therapeutic strategies for this high-risk phenotype.