Background: In the recent STEP-HFpEF program, high dose treatment with the GLP-1 receptor agonist semaglutide demonstrated an improvement in heart failure (HF)-related symptoms, a reduction of NT-proBNP levels and acute HF hospitalizations in HFpEF patients across multiple studies. Intriguingly, treatment effects were independent of baseline body weight and left-ventricular ejection fraction. Moreover, patients with higher NT-proBNP at baseline exhibited more pronounced improvement in HF symptoms despite similar weight loss. These aspects spark interest regarding direct cardiac effects of semaglutide.
Purpose: We tested the effects of acute semaglutide treatment reflecting clinically relevant concentrations in isolated human atrial myocardium and cardiomyocytes.
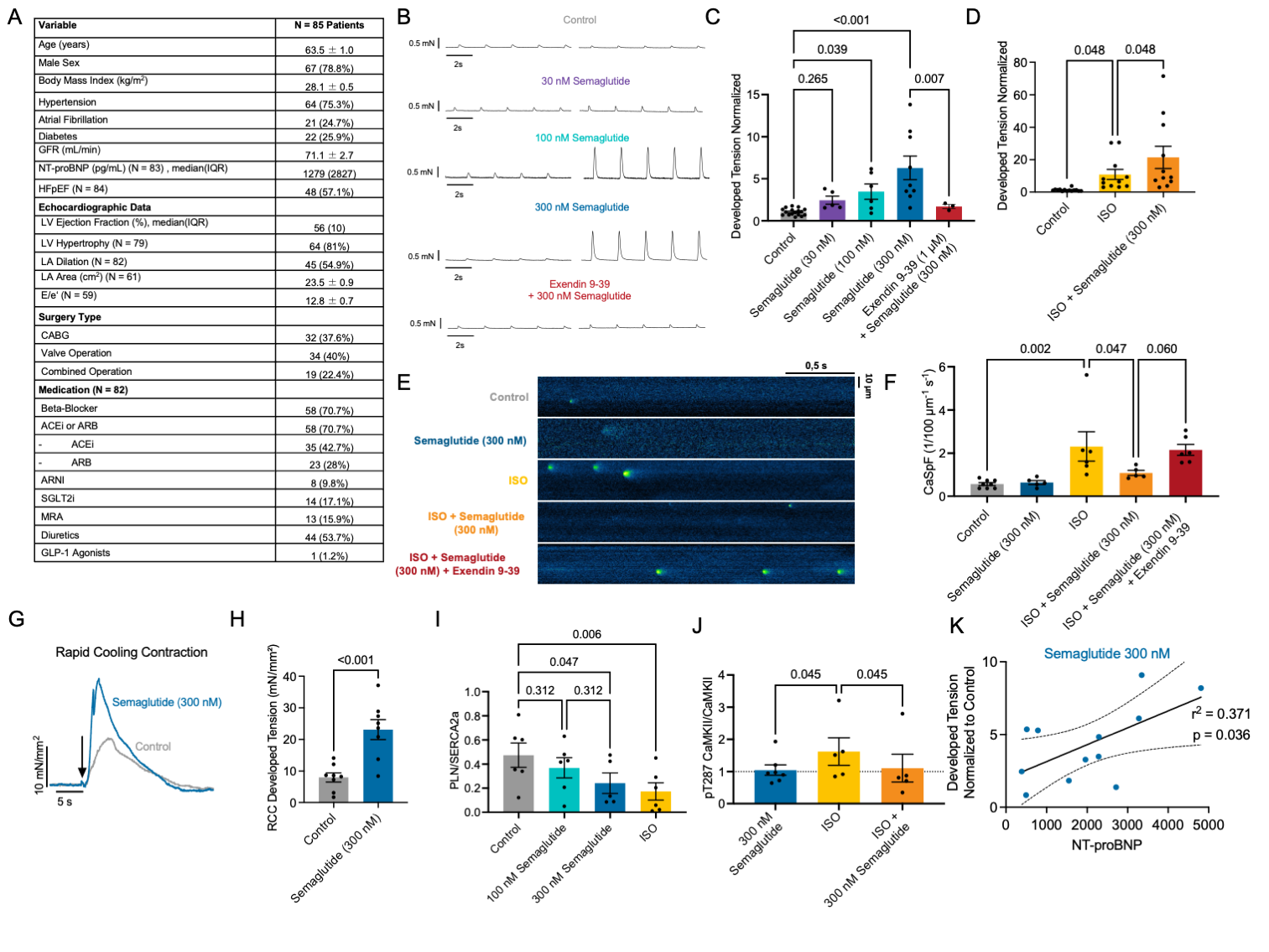
Methods and Results: Atrial myocardium and clinical data was analyzed from 85 patients undergoing elective coronary artery bypass grafting and/or valve surgery. Atrial trabeculae and myocardial slices were prepared from right-atrial appendage biopsies. Regular contractions were elicited by electrical field stimulation (0,5 - 1Hz, 37°C). Where possible, paired experiments were conducted with control and treated groups from the same patient. Most patients exhibited HFpEF according to current guideline criteria (Figure A). Acute exposure of myocardial slices to semaglutide resulted in a concentration-dependent increase in developed tension (Figure B+C). Blockade of the GLP-1 receptor (Exendin 9-39) confirmed receptor dependence. Semaglutide treatment also resulted in a superimposed positive inotropic effect on top of beta-adrenergic stimulation with isoprenaline (Figure D). In isolated human atrial cardiomyocytes, baseline diastolic Ca spark frequency was low, but when increased by beta-adrenergic stimulation, additional semaglutide treatment reduced Ca spark frequency profoundly (Figure E+F). This effect was also reversed by GLP-1 receptor antagonism with Exendin 9-39. In atrial trabeculae, semaglutide also increased SR Ca content as indirectly assessed by rapid cooling contractions (Figure G+H). Mechanistically, phospholamban/SERCA2a ratio assessed by Western blot was reduced only by high concentrations of semaglutide (I) hinting at increased SR Ca re-uptake. When CaMKII was stimulated by beta-adrenergic activation, treatment with semaglutide (300 nM) rescued autophosphorylated pT287 CaMKII /total CaMKII ratio (immunohistochemistry) almost back to baseline levels (J), congruent with the diastolic Ca spark analysis. This provides evidence for potential cardioprotective effects of semaglutide in atrial myocardium especially when pathological stimuli/ remodeling are present. An exploratory comparison of the improvement in contractility resulting upon semaglutide exposure with patient NTproBNP levels revealed a positive correlation in linear regression analysis (K), underscoring the effects observed especially in patients with higher NT-proBNP values in the STEP-HFpEF program.
Conclusion: In summary, semaglutide induces a GLP-1 receptor–dependent improvement in atrial contractility via enhanced SR Ca handling and reduced PLN/SERCA2a ratio, without promoting arrhythmias and detrimental CaMKII activation. These findings provide groundwork for more in-depth investigation into semaglutide as a therapeutic agent in HFpEF.