Introduction:
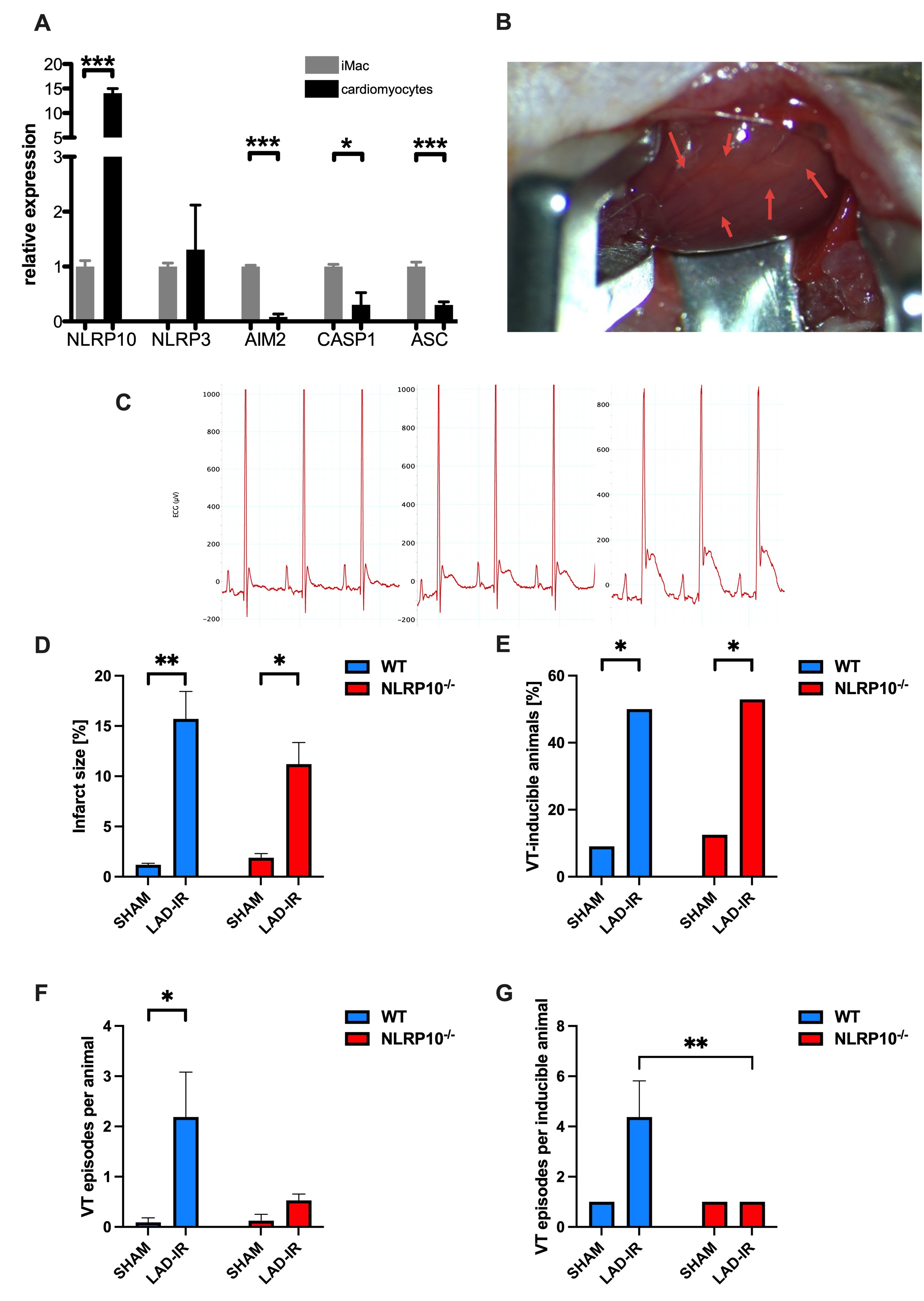
The cytosolic pattern recognition receptor NLRP10 has been implicated in regulating innate immune and inflammasome responses, particularly following mitochondrial damage. Its role in post-ischemic cardiac remodeling and arrhythmogenesis remains largely unexplored. Cardiomyocytes, characterized by a high mitochondrial density to meet energy demand, also exhibit strong NLRP10 expression (A). We hypothesized that NLRP10 deficiency affects inflammatory and electrophysiological remodeling after myocardial infarction (MI), thereby influencing the progression toward ischemic cardiomyopathy (ICM).
Methods:
NLRP10-deficient (NLRP10-/-) and wild-type (WT) mice underwent transient occlusion of the left anterior descending coronary artery followed by reperfusion (LAD-IR) using a minimally invasive “closed-chest” model (B). ECG tracings confirmed successful induction of myocardial ischemia during LAD-IR (C: representative recordings at baseline, acute occlusion, after 1 hour). Cardiac function was evaluated by echocardiography at baseline and 14 days post-intervention. Histological analyses quantified infarct size and interstitial fibrosis. Invasive electrophysiological studies were performed to determine conduction parameters and arrhythmia susceptibility. Sham-operated animals served as controls.
Results:
At baseline, cardiac morphology and function did not differ between genotypes. LAD-IR resulted in a marked decline of left ventricular ejection fraction (WT: 49.1 ± 1.6 % vs. 30.3 ± 3.2 %, p ≤ 0.01; NLRP10-/-: 52.4 ± 3.2 % vs. 34.5 ± 4.1 %, p < 0.05), without differences between groups (p = 0.6). Infarct size (D) and inflammatory cell infiltration were comparable. ECG analyses demonstrated QT and QTc prolongation after LAD-IR in both groups, indicating increased repolarization heterogeneity. Notably, NLRP10-/- mice displayed significantly shorter ventricular refractory periods (WT LAD-IR: 32.2 ± 2.5 ms vs. NLRP10-/- LAD-IR: 25.0 ± 1.1 ms, p ≤ 0.01). During ventricular stimulation, inducibility of ventricular tachycardia (VT) was comparable between groups (E; WT: 50.0 %; NLRP10-/-: 52.9 %). However, the increase in VT episodes per animal observed after LAD-IR in WT was absent in NLRP10 deficiency (F). The number of VT episodes per inducible animal was markedly lower in NLRP10-/- mice (G: WT 4.4 ± 1.4 vs. NLRP10-/- 1.0 ± 0.0, p ≤ 0.05). Likewise, atrial stimulation showed fewer AF episodes per inducible NLRP10-/- animal, while overall AF inducibility remained comparable.
Conclusion:
NLRP10 deficiency does not alter cardiac morphology or systolic function after ischemia-reperfusion but nevertheless seems to provide protection against ventricular tachyarrhythmia. This study analyses immune-electrical interactions in post-ischemic hearts and highlights NLRP10 as a potential modulator of electrophysiological stability during ventricular remodeling. Further mechanistic studies are warranted to define the molecular pathways linking NLRP10 signaling, mitochondrial stress, and electrophysiological stability.