Background: Cardiac arrhythmias are highly prevalent in aging populations, but biological mechanisms linking aging and arrhythmogenesis remain poorly understood. Clonal hematopoiesis of indeterminate potential (CHIP) and its cytopenic variant, clonal cytopenia of undetermined significance (CCUS), are age-related precursor states that can drive systemic inflammation and myocardial remodeling. While CHIP has been linked to cardiovascular disease, data on CCUS in patients with heart rhythm disorders are virtually absent.
Purpose: To determine the prevalence of cytopenia and CCUS among patients with cardiac arrhythmias and explore associations with arrhythmia subtype and prognosis. Methods: We screened 2,066 consecutive adults treated at a tertiary rhythmology center (2021–2025). After systematic exclusion of secondary causes, patients with unexplained cytopenia (Hb < 10 g/dL, leukocytes < 2×109/L, or platelets < 150×109/L) underwent targeted next-generation sequencing (NGS) of 159 myeloid genes. CCUS was defined as ≥ 1 pathogenic or likely pathogenic variant (variant allele frequency (VAF) ≥ 1%) in a CHIP-associated gene without hematologic malignancy. Prognostic validation was performed in matched real-world cohorts using the TriNetX Global Health Research Network.
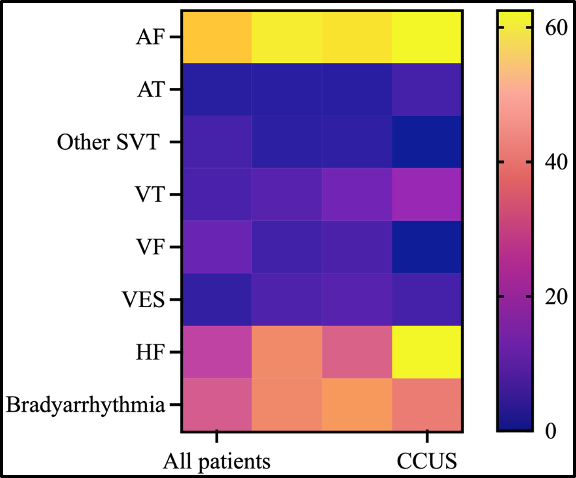
Results: Among 1,983 patients with laboratory data, cytopenia was found in 42.0%, and unexplained cytopenia in 10.2% (202/1,983). Rates were highest in heart failure (16.8%) and ventricular tachycardia (15.2%), and lowest in supraventricular tachycardia (3.4%) (Figure1). Individuals with unexplained cytopenia were older (median 82 years) and predominantly male (71.8%). Cytopenia was associated with a 1.27-fold higher risk of persistent/permanent AF (p = 0.0021). In > 500,000 matched patients within TriNetX, any cytopenia predicted significantly reduced overall survival across arrhythmia subtypes (log-rank p < 0.001).
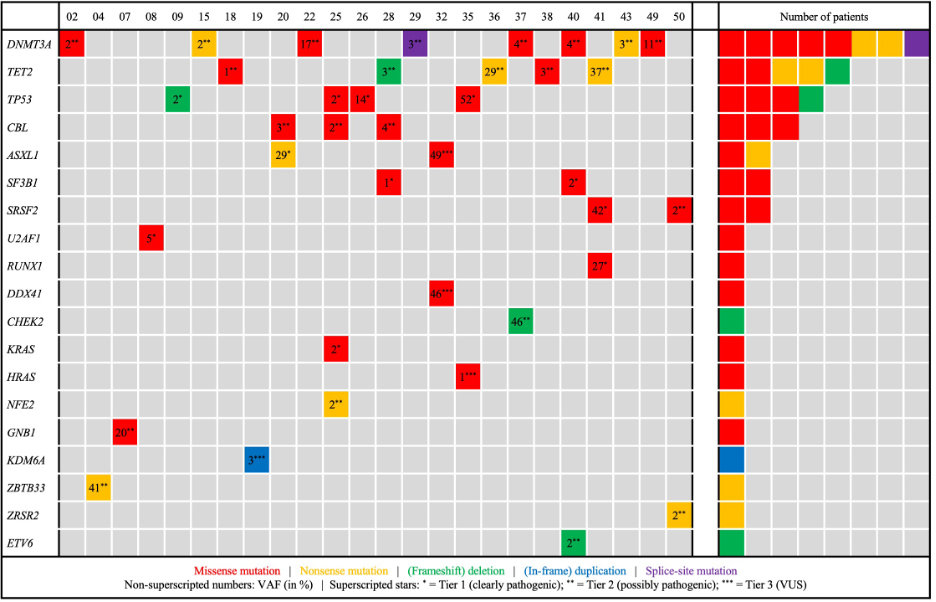
Of 49 patients sequenced, 24 (49.0%) met diagnostic criteria for CCUS, corresponding to an estimated overall CCUS prevalence of ~5% in the entire arrhythmia cohort. The most frequent driver genes were DNMT3A (33.3%), TET2 (20.8%), TP53 (16.7%), and spliceosome genes (20.8%); 41.7% of patients carried multiple mutations (Figure 2). CCUS was enriched in atrial fibrillation (54.5%) and heart failure (55.6%) (Figure 1).
Conclusion: Unexplained cytopenia and CCUS are frequent but under-recognized in rhythmology patients, particularly in older men with atrial fibrillation or heart failure. This study provides the first systematic evidence of CCUS in arrhythmia populations, suggesting a potential inflammatory and clonal hematopoietic substrate for arrhythmogenesis. Routine blood count screening and hematology referral could improve risk stratification and identify candidates for anti-inflammatory or clonal-directed therapy.