BackgroundIdentification of acute coronary occlusion (ACO) in chest pain patients with wide QRS complexes remains clinically challenging. While several ECG criteria exist for left bundle branch block (LBBB) or ventricular pacing (VP), their complexity limits clinical use. For right bundle branch block (RBBB) or non-specific intraventricular conduction delay (NIVCD), no standardized ECG criteria are established. Artificial intelligence–enhanced ECG analysis may improve ACO detection, though its value in these subgroups remains unclear.
ObjectiveTo assess the diagnostic performance of an AI-enhanced ECG model for detecting ACO in chest pain patients with wide QRS complexes in a metropolitan chest pain unit (CPU) cohort. In LBBB and VP subgroups, its performance was compared to established ECG criteria.
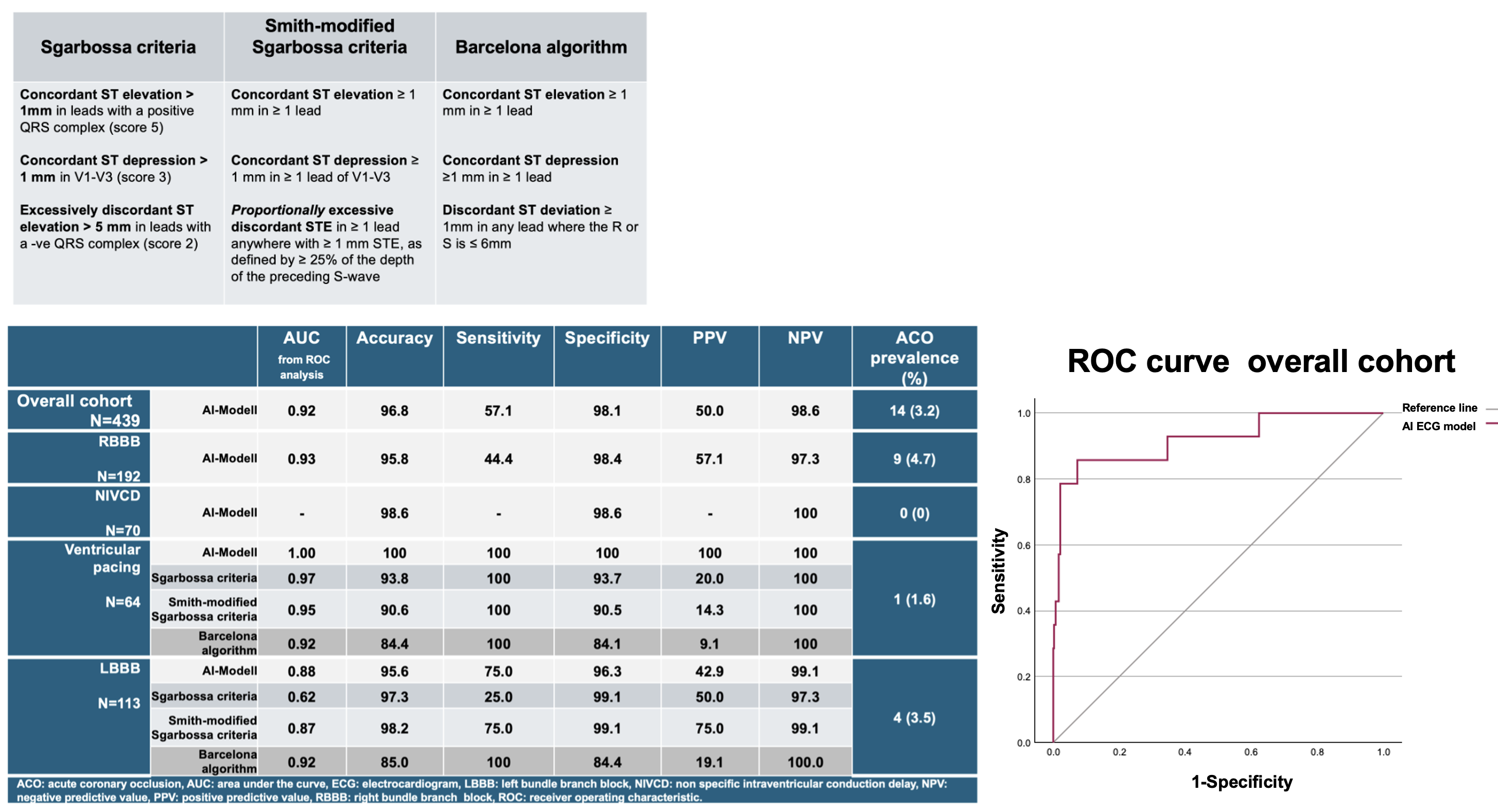
MethodsThe AI model, built on a deep convolutional neural network, had been trained and externally validated in international cohorts. For this analysis, all consecutive chest pain patients presenting to the CPU between November 2018 and June 2021 with a QRS duration ≥120ms were included. ECGs were independently reviewed and classified by two trained investigators; discrepancies were resolved by a third author. Clinical management followed current European Society of Cardiology recommendations, including the 0/1-hour high-sensitivity cardiac troponin T algorithm for low risk rule-out. The endpoint was ACO, adjudicated by an independent physician blinded to the AI model’s predictions integrating coronary angiography, biomarkers, and echocardiography. Diagnostic performance was evaluated using receiver operating characteristic (ROC) analysis. In LBBB and VP subgroups, AI metrics were compared to Sgarbossa, Smith-modified Sgarbossa, and Barcelona criteria (Table 1).
ResultsAmong 4,440 CPU admissions, 439 patients with wide QRS complexes were included: 192 (43.7%) RBBB, 113 (25.7%) LBBB, 70 (15.9%) NIVCD, and 64 VP (14.6%). Median QRS duration was 144ms [132-158]. ACO prevalence was 3.2% (n = 14: 9 RBBB, 4 LBBB, 1 VP and 0 NIVCD).
Diagnostic metrics are summarized in Table 2 and Figure 1. Overall, the AI-ECG model detected 8 of 14 ACO cases (57%). In the low-risk rule-out population (n=216), the AI-ECG model yielded two false positives, potentially indicating two unnecessary emergency angiographies.
In RBBB and NIVCD, AI-ECG model showed remarkable specificity, while showing an acceptable sensitivity in RBBB. In VP patients, AI-ECG model classified all cases correctly and outperformed conventional ECG criteria by avoiding false positives (PPV 100% vs. 9.1-20%).
In LBBB patients, Barcelona algorithm, and Smith-modified Sgarbossa criteria all demonstrated excellent diagnostic metrics. Compared to Barcelona criteria, the AI-ECG model reduced false positives again.
ConclusionThe AI-ECG model demonstrated high specificity in RBBB, where no established ECG reference standard exists, supporting further evaluation in independent cohorts. In chest pain patients with LBBB, it achieved diagnostic accuracy comparable to established but complex ECG criteria, while outperforming them in ventricular pacing. Pending external validation in cohorts with higher ACO prevalence, it may offer a rapid and simple diagnostic alternative.