Background: The mechanistic target of rapamycin complex 1 (mTORC1) is a central regulator of cellular metabolism, growth, and autophagy that is essential for the maintenance of cellular homeostasis in cardiomyocytes. Chronic mTORC1 hyperactivation promotes maladaptive remodelling, hypertrophy, and heart failure. TSC2 is a crucial upstream inhibitor of mTORC1 that integrates metabolic and mechanical stress signals. Phosphorylation of TSC2 at Serine 1364/1365 has been proposed as a stress-sensitive regulatory mechanism to modulate mTORC1, but its functional role in human cardiomyocytes is undefined.
Methods: Human induced pluripotent stem cell (hiPSC) were generated expressing wild-type TSC2, a phospho-mimetic TSC2SE (Ser→Glu), or a phospho-dead TSC2SA (Ser→Ala). Cells were differentiated into cardiomyocytes (hiPSC-CMs) and cultured under standardized maintenance conditions. Cardiomyocyte purity and maturation were confirmed both by immunofluorescence and flow cytometry. For functional assays, cells were treated for 24 hours with 200 µM palmitate or 7.1 mM lactate in 5.5 mM glucose medium, or high glucose medium before measurements. Contractility was assessed by high-speed motion analysis, while calcium transients were examined by loading FURA-2 AM (30 minutes at 37 °C) and calcium imaging (IonOptix).
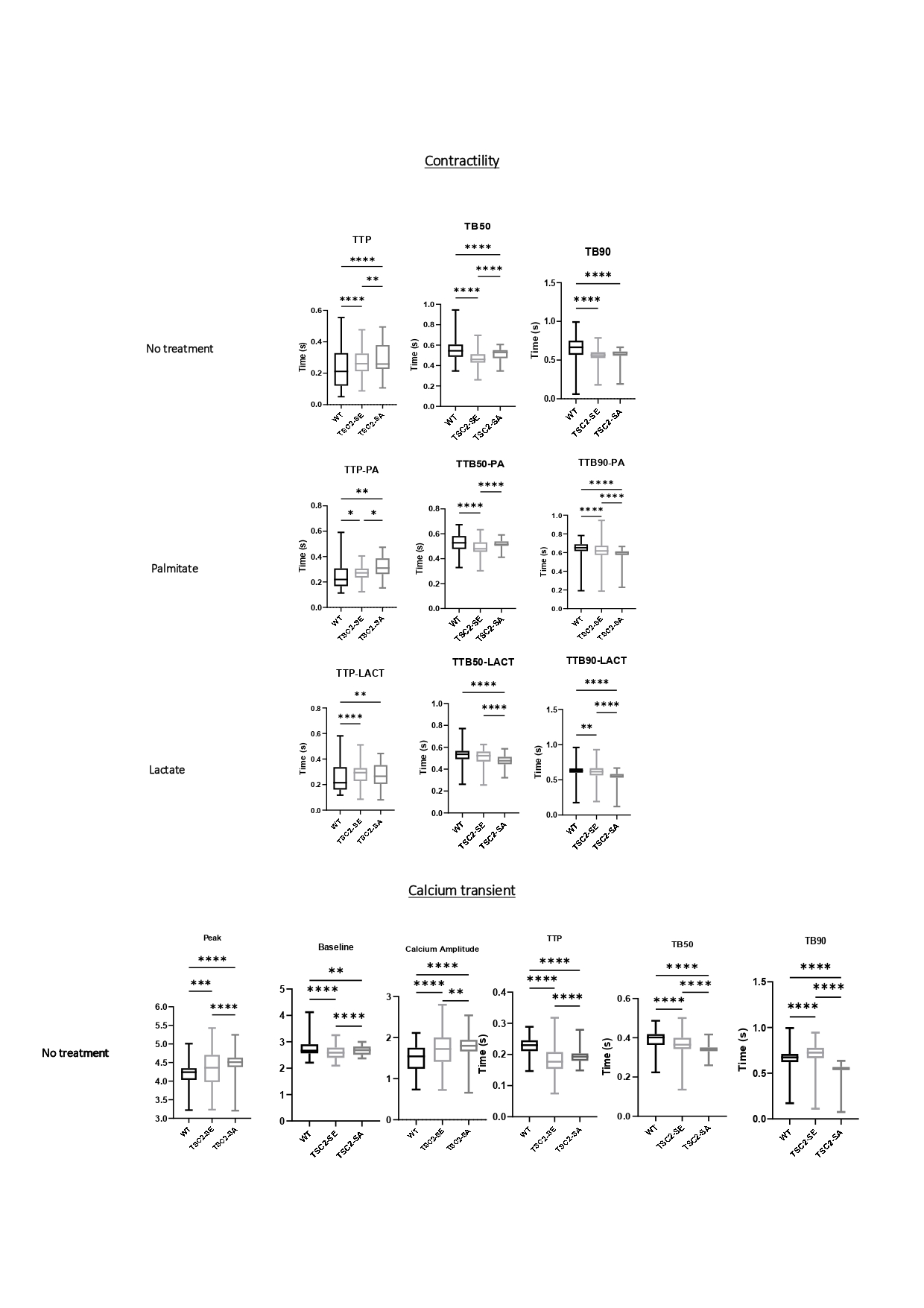
Results: TSC2SA cardiomyocytes demonstrated a hypercontractile phenotype, with uniformly shorter contraction and relaxation times across all conditions tested as well as increased calcium transient amplitude compared to TSC2WT and TSC2SE hiPSC-CMs. In TSC2SE cells, treatment with palmitate and lactate decreased calcium transient amplitude but hastened contraction and relaxation times, demonstrating a dissociation between the magnitude of the calcium signal and mechanical output. This suggests a critical role for TSC2 phosphorylation in modulating excitation-contraction coupling through increased myofilament Ca²⁺ sensitivity, accelerated cytosolic Ca²⁺ clearance, or post-translational modifications of contractile proteins. These data establish that the phospho-dead TSC2SA knock-in mutant, and more broadly TSC2 phosphorylation state, controls mTORC1-dependent cardiomyocyte contractility and functional adaptation to stress.
Conclusion: This study provides experimental evidence linking TSC2S1364/1365 phosphorylation with cardiomyocyte contractile performance. The hypercontractile phenotype observed in all conditions for TSC2SA cells and the perturbations in calcium dynamics in TSC2SE cells underpin a phosphorylation-dependent mechanism controlling mTOR signalling, further providing potential therapeutic targets to restore mTOR homeostasis and abrogate maladaptive remodelling in heart failure.
Keywords: mTOR; TSC2; phosphorylation; hiPSC-cardiomyocytes; contractility; cardiac remodelling; heart failure.