Background
High sensitivity cardiac troponins (hs-cTn) are the gold standard biomarkers in diagnosing acute myocardial infarction (AMI). Chronic myocardial stress from atrial fibrillation (AF) or impaired renal function can lead to troponin elevation to challenges in diagnosis of AMI with classical troponin-based models.Explainable Boosting Machines (EBMs) are interpretable additive models that use gradient boosting to learn a distinct shape function for each feature, maintaining interpretability while capturing non-linear relationships.This study aimed to assess if explainable machine learning (ML) models may assist in better interpretation of the interaction of hs-cTn with comorbidities in patients with suspected AMI.
Methods
Data from a large multi-centric cohort that enrolled patients with suspected AMI were analyzed.The adjudicated AMI diagnosis according to the universal myocardial infarction definition was the outcome of interest. Patients were grouped according to the diagnosis of ongoing AF using 12-lead ECG or according to presence of impaired renal function defined as a glomerular filtration rate (GFR) <45mL/min/1.73m² at presentation. EBM models were trained separately for each subgroup using 80/20 stratified train-test splits. Shape functions were extracted from the trained EBM models to visualize the relationship between feature values and AMI risk. Each function shows how feature’s values affect AMI risk on the log-odds scale, and the final prediction is the sum of all feature contributions plus an intercept. This additive structure enables direct interpretation of how individual features contribute to AMI risk, allowing direct visualization of clinically relevant thresholds and non-linear relationships.
Results
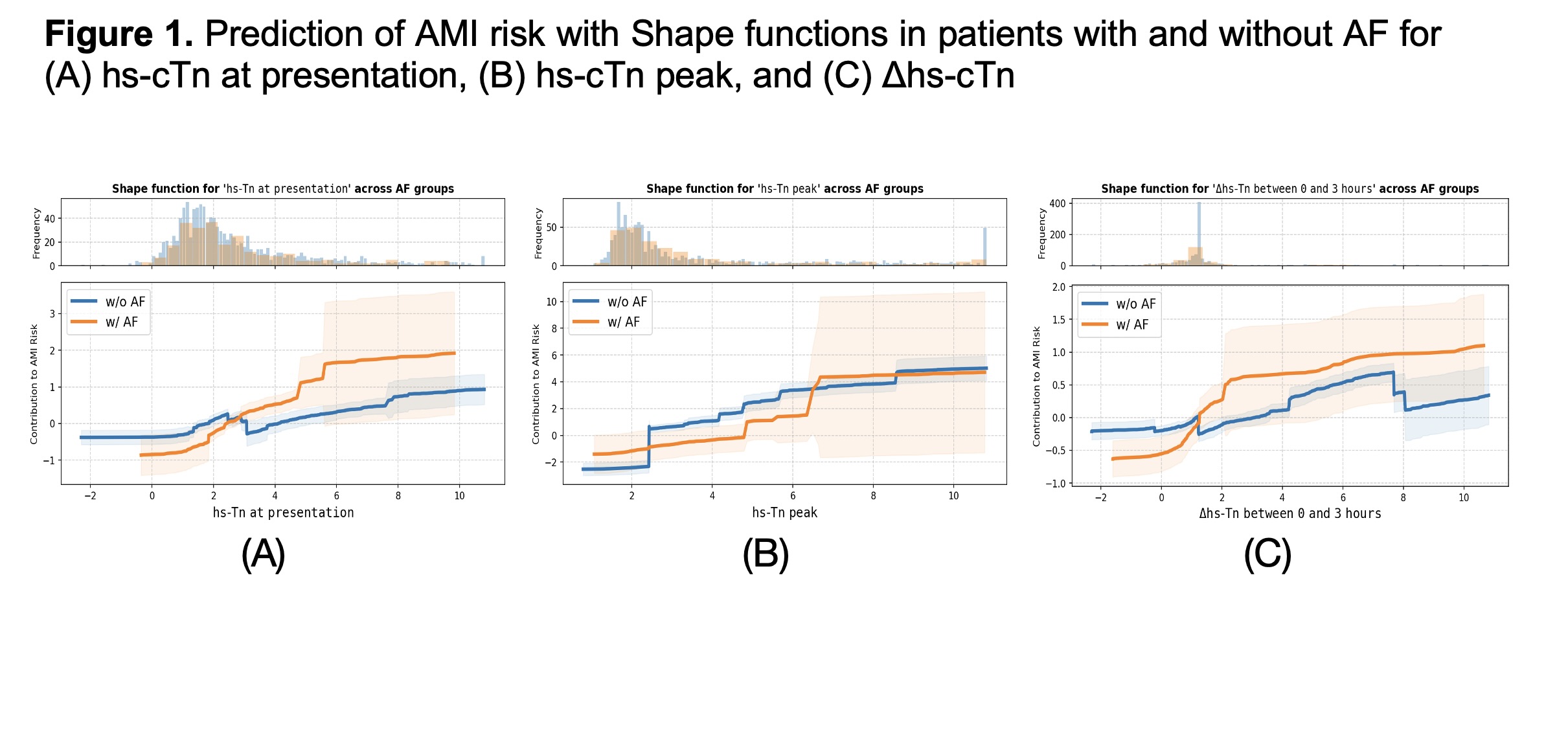
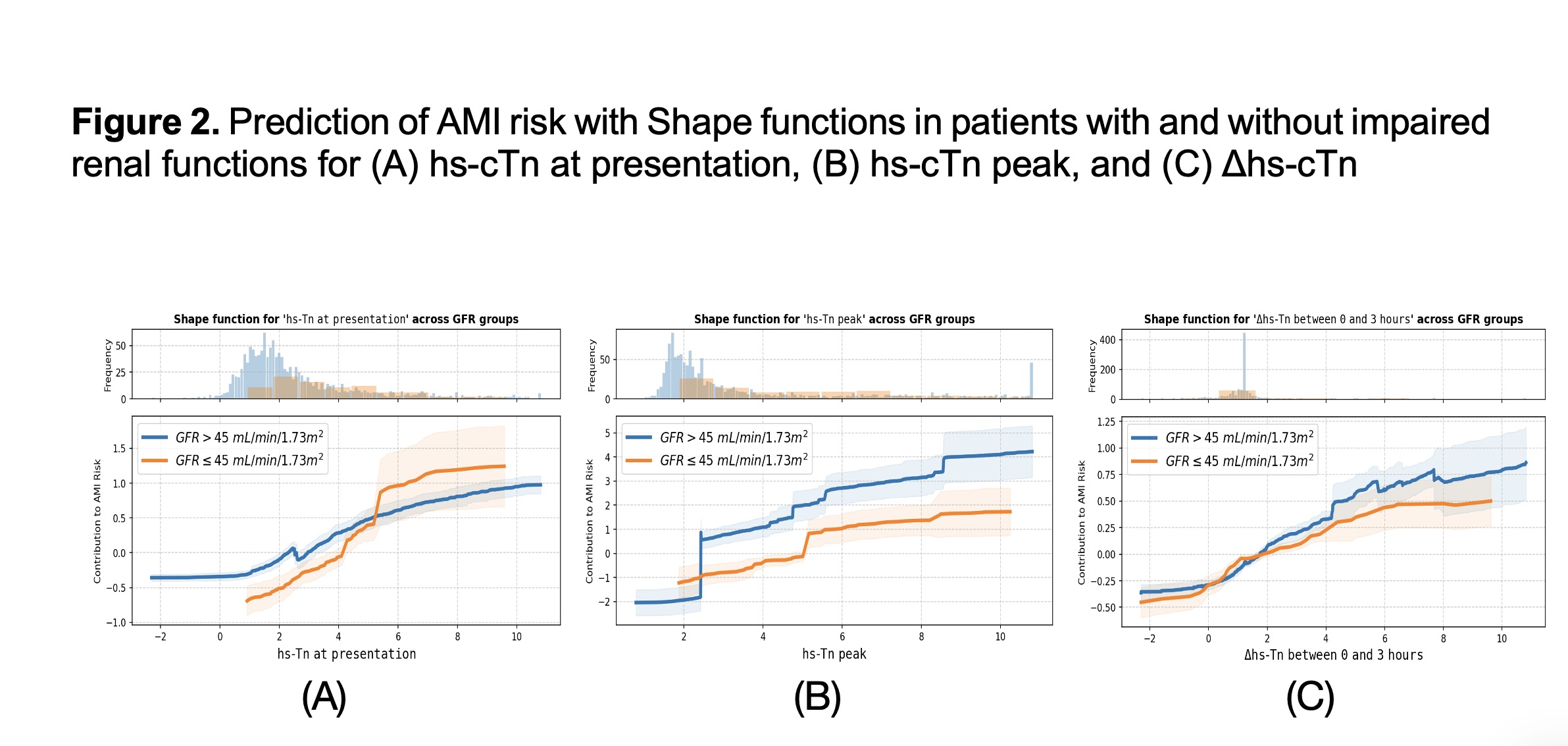
We included 1818 patients with a median age of 61.4 (standard deviation, SD 13.5) years and 1208 (66.4%) were males. AF was present in 314 (17.2%) patients and mean glomerular filtration rate (GFR) was 79.0 (SD 21.4). A total of 413 (22.7%) patients had acute AMI event. In patients with AF, increased hs-cTn at presentation contributed more strongly to the prediction of AMI in patients with ongoing AF compared with those without AF (Figure 1A), whereas peak hs-cTn values had a more consistent impact suggesting a less discriminative effect in AF patients (Figure 1B). Notably, the threshold of baseline hs-cTn for predicting AMI was higher in AF-patients. Dynamic hs-cTn changes within 3h after presentation contributed more strongly to the AMI prediction in AF patients than non-AF ones (Figure 1C). In patients with impaired renal function, hs-cTn at presentation predicted acute AMI strongly only at high thresholds (Figure 2A). In contrast, peak hs-cTn showed higher predictive capacity in patients with impaired renal function (Figure 2B), while dynamic hs-cTn changes had a consistent effect (Figure 2C).
Conclusions
In patients with suspected AMI applying an explainable ML model can facilitate the understanding of the interactions and diagnostic capacity of hs-cTn in patients with AF or impaired renal function. At a higher threshold, baseline hs-cTn values had better predictive capacity for AMI in patients with AF and impaired renal function.
