Context and Purpose
Chronic kidney disease (CKD) affects around 10% of the global population and markedly increases cardiovascular morbidity and mortality. The uremic toxin indoxylsulfate (IS) disrupts myocardial calcium (Ca²⁺) homeostasis and promotes arrhythmias. We recently showed that SGLT2 inhibitors (SGLT2i) improve myocardial Ca²⁺ handling and prevent cardiac arrhythmias. This study investigates the anti-arrhythmic effect of SGLT2i in a CKD model.
Methods
Isolated murine ventricular cardiomyocytes were loaded with Fluo-4 and Ca²⁺ release events were measured by confocal microscopy as a measure of Ca²⁺ handling and arrhythmogenicity. Uremia was simulated by 120 mg/l indoxyl sulfate (IS), reflecting peak plasma levels in dialysis patients. Human induced pluripotent stem cell-derived cardiomyocytes (iPSC-CM) were exposed to IS for 15 min or 7 days, followed by analysis of Ca²⁺ spark frequency (CaSpF) and Ca²⁺ amplitude. Selected experiments were performed in SGLT2-knockout mice (SGLT2-KO), and intervention was done by SGLT2i empagliflozin (EMPA, 1 µM), dapagliflozin (DAPA, 1 µM), or the CaMKII inhibitor AIP (1µM). For reactive oxygen species (ROS) measurements, murine cardiomyocytes were stained with CellROX Orange and perfused with IS+EMPA for 15 min. Statistical analyses were performed with Kruskal–Wallis and two-way ANOVA tests.
Results
In all experimental settings, IS increased CaSpF (SGLT2-KO: 0.2 (0.4) vs. 0.5 (0.8); acute iPSC-CM model: 0.5 (0.8) vs. 1.2 (1.9); chronic iPSC-CM model: 0.5 (0.7) vs. 1.0 (1.0)), indicating a pro-arrhythmic effect. Co-incubation with EMPA significantly reduced CaSpF in murine cardiomyocytes and in iPSC-CMs (SGLT2-KO: 0.5 (0.8) vs. 0.2 (0.4); acute iPSC-CM model: 1.2 (1.9) vs. 0.3 (0.5); chronic iPSC-CM model: 1.0 (1.0) vs. 0.4 (0.6)). All effects were observed following both acute and chronic incubation. In SGLT2-KO cells, DAPA and AIP also reduced the CaSpF. Incubation with EMPA, DAPA and AIP alone did not alter CaSpF. IS did not affect Ca²⁺ amplitude (data not shown). Perfusion with IS increased ROS levels, whereas co-application of EMPA exerted a protective effect, suggesting a protective mechanism linked to its antiarrhythmic effect.
Conclusion
Beyond confirming the proarrhythmogenic effects of IS, our data suggest a protective cardiac role of SGLT2i in the context of CKD. Since this effect was shown in SGLT2-KO cardiomyocytes it should be independent of SGLT2 expression. The comparable effects between AIP and SGLT2i support the hypothesis that SGLT2i may confer antiarrhythmic effects by targeting the CaMKII signaling pathway. The observed pharmacological modulation offers a potential therapeutic strategy to mitigate cardiovascular risk in CKD patients.
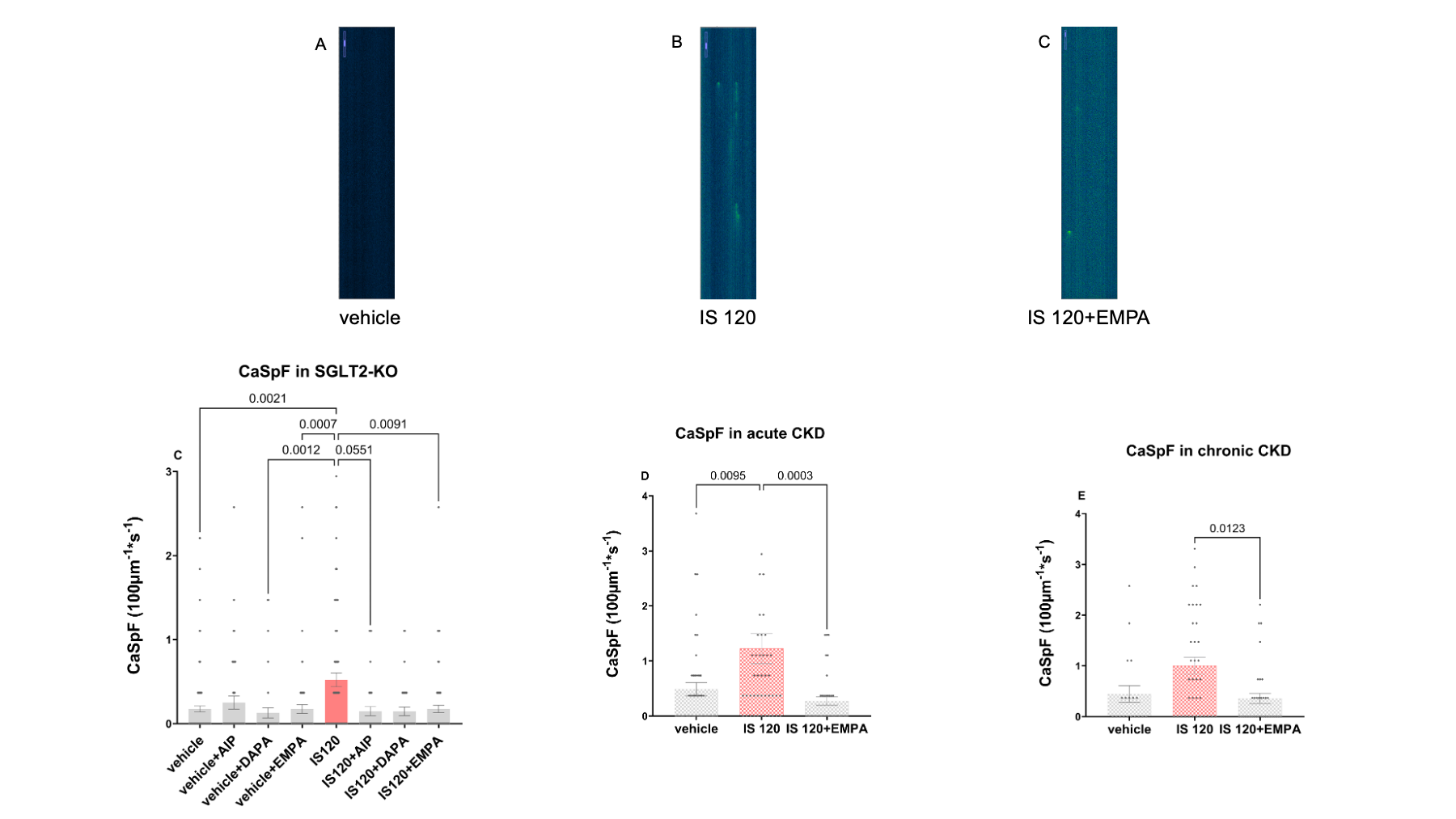
Figure 1 Confocal measurements showed an increased CaSpF for 15 minutes incubation with IS 120 mg/L in SGLT2-KO cells and a reduction in co-incubation with EMPA, DAPA and AIP. Incubation with DAPA, EMPA, AIP alone did not show any effect (C). iPSC-CM measurements showed a similar effect both for 15 minutes incubation (D) and for 7 days incubation (E) with Indoxylsulfat and EMPA.
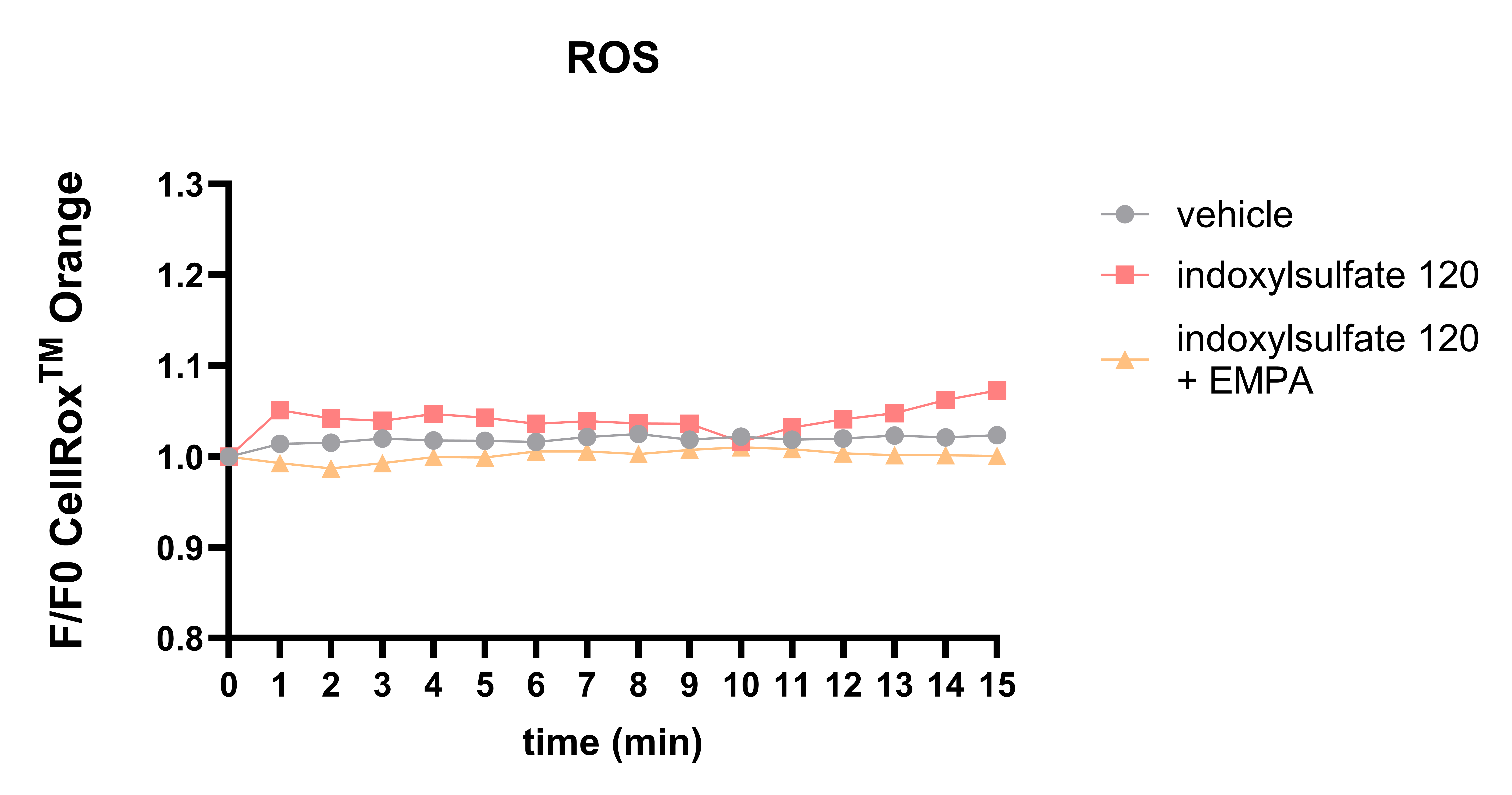
Figure 2 Perfusion with IS120 led to an elevation of ROS, whereas co-incubation with IS and EMPA highlighted the protective effects of SGLT2i.
