Objective: Adherence is a fundamental component of medical therapy. This study aimed to determine medication adherence in patients with pulmonary arterial hypertension (PAH) by assessing patient questionnaires and plasma drug concentrations.
Methods: This single-centre, retrospective and partially cross-sectional study involved the routine assessment of adherence in patients with PAH who presented at our centre between December 2024 and October 2025. Adherence was measured using the A14 self-report questionnaire and plasma levels of common PAH medications (phosphodiesterase type 5 inhibitors (PDE5i) and endothelin receptor antagonists (ERA)). The A14 scale classifies patients as adherent if they score between 50 and 56, or as non-adherent if they score lower than 50. Drug levels were analysed according to the multiple of the expected mean plasma drug concentration (MoM).
Results: The study includes 249 patients (mean age 66 years; 67.3% female; 58.3% idiopathic PAH; 4.7% heritable PAH; 5% PAH associated with congenital heart disease; 18% PAH associated with connective tissue disease) taking oral PAH medication > 3 months. The A14 scale classified 27 patients (10.8%) as non-adherent and 222 patients (89.2%) as adherent. A plasma concentration lower than 0.5 MoM, defined as an insufficient drug level, was detected in 23.9% of patients receiving PDE5i with already determined drug levels and in 10.2% receiving ERA with already determined drug levels (Figure 1).
Conclusions: This study is the first to confirm that a large proportion of PAH patients receiving PDE5i and/or ERA have inusfficient plasma concentrations, indicating an ineffective dose. This could be due to lower adherence, as measured by the A14 scale, or to individual pharmacokinetics, metabolism, or possible medication interactions. Thus, reduced adherence and consequently reduced drug levels are major problems in the treatment of PAH that cannot be detected by questionnaires alone.
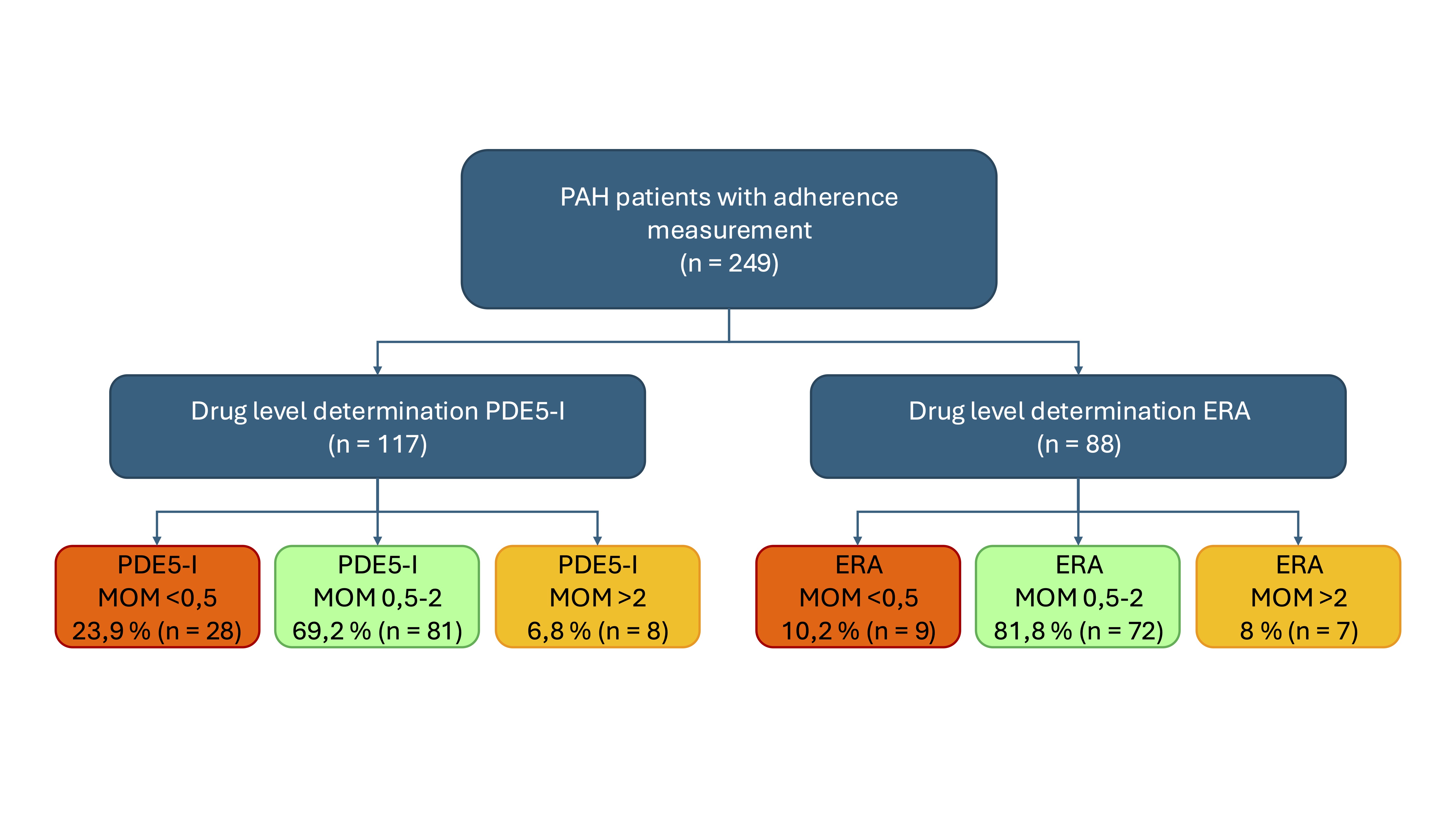
Figure 1. Study flow chart: Current status of drug level measurement. Some drug levels are not yet listed here as they are still being evaluated. It’s possible that patients take both classes of active substances and can be classified in both groups. The completion and evaluation of drug level measurements will follow in the course of the study.
