Takotsubo syndrome (TTS) is a reversible form of heart failure leading to regional ventricular contraction irregularities. It typically affects postmenopausal women following a stressful trigger. TTS is characterized by a transient ballooning of the left ventricle during systole, likely due to apical hypocontractility, combined with hypercontractility of the basal segments. This ballooning is typically accompanied by a rightward shift of the pressure-volume-loop (PV loop). The underlying pathophysiology of TTS remains unclear, with possible explanations being a corruption of the Purkinje-muscle junctions (PMJs) in the apical region or change in active stress development due to sympathetic overdrive.
We hypothesized three different mechanisms that could reproduce the ballooning and tested them in a computational electromechanical heart model. In all three scenarios we divided the ventricles in an apical region and a basal region. For scenario A, we used a heart model with an integrated His-Purkinje system and deactivated the PMJs in the apical region while leaving conduction and stress development in the myocardium intact. In scenario B, we disabled the active stress development in the apical region to simulate deactivation or lack of beta-adrenergic receptors. For scenario C, we modified scenario B by additionally doubling the active stress development in the basal region of the ventricles. Results were compared against a control simulation without any alterations.
The deactivation of the PMJs led to a slightly delayed onset of contraction in the apex but showed no ballooning at the time of full contraction (Fig. 1A). There was no change in left ventricular ejection fraction (LVEF) compared to the control simulation. The disabled active stress development in the apex (case B) resulted in a visible ballooning effect (Fig. 1B). Case B also presented with a decreased LVEF (37.45%) compared to control (56.59%) and a rightward shift of the PV loop. The ballooning was even more pronounced for the third simulation scenario (Fig. 1C). This case also demonstrated a decrease in LVEF (43.91%) and a rightward shift of the PV loop, although not as pronounced as for case B.
Our findings show that a corruption of the PMJs cannot be the sole cause for the TTS phenotype. According to scenarios B and C, the hypocontractility in the apical region appears as the dominant mechanism contributing to apical ballooning. Furthermore, the hypercontractility close to the base, modeled in case C, leads to a partial recovery of pump function and demonstrates an even more characteristic ballooning contraction pattern. Thus, mid/basal hypercontractility could be another contributing mechanism. The role of the sympathetic nervous system on active stress development and apical ballooning warrants exploration in follow-up work.
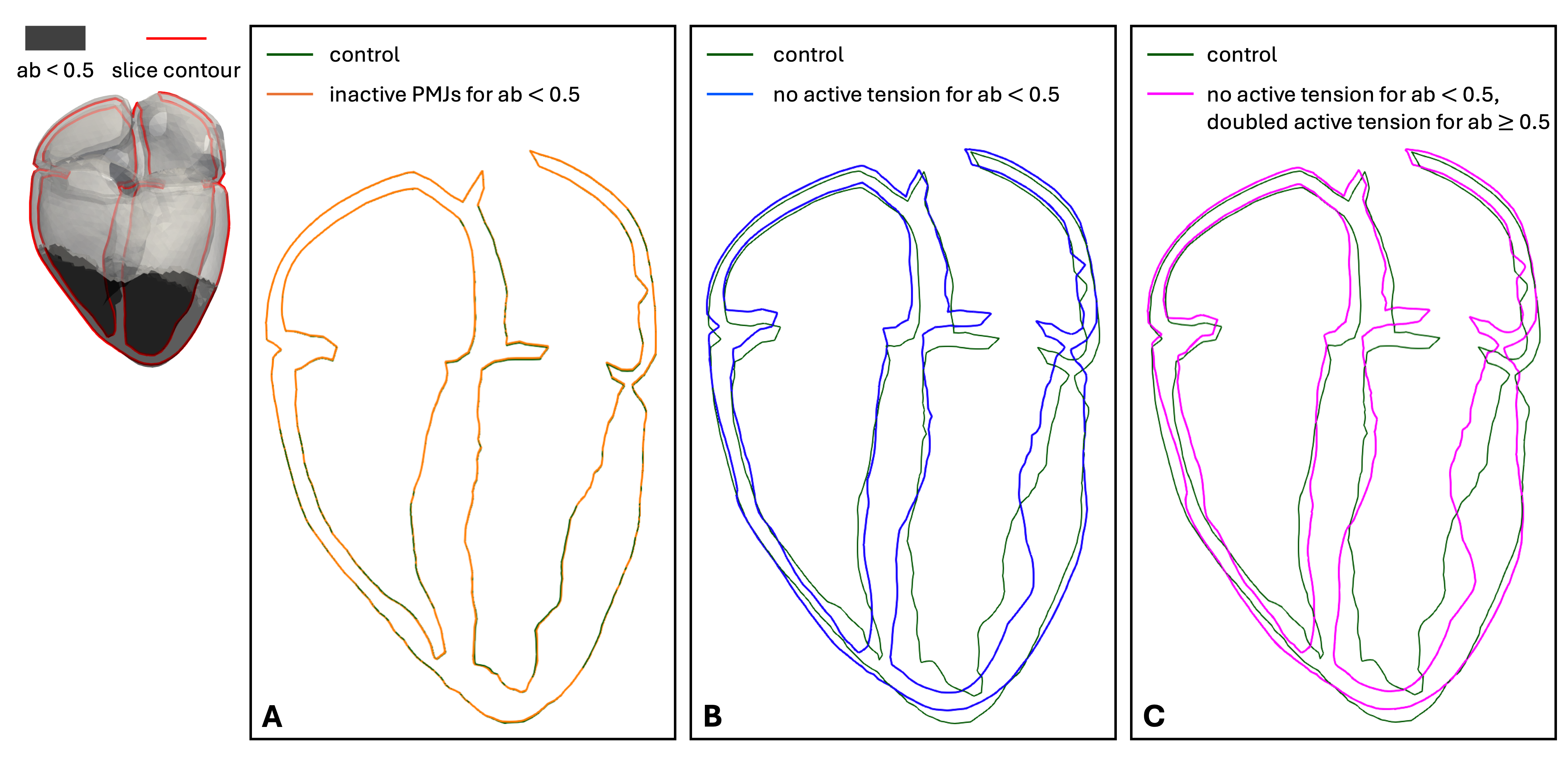
Figure 1: 4-chamber slice of all simulations at the time of full contraction. Top left: apical region (apico-basal (ab) coordinate < 0.5) and the position of the slice. The different simulations are depicted in different colors. PMJs: Purkinje-muscle junctions.
