Background Transthyretin amyloid cardiomyopathy (ATTR-CM) is a progressive form of restrictive cardiomyopathy caused by the deposition of misfolded transthyretin (TTR) in the myocardium, leading to heart failure. While amyloid deposition is central to disease pathology, the contribution of immune-mediated mechanisms—particularly neutrophil activation and formation of neutrophil extracellular traps (NETs)—remains poorly understood. NETs and NLRP3 inflammasome activation have been implicated in chronic inflammation and tissue remodeling, suggesting a potential role in ATTR-CM pathogenesis
Aims This study aimed to investigate the extent and spatial distribution of neutrophil activation and NET deposition in cardiac tissue of ATTR-CM patients, to evaluate the relationship between neutrophil activation and amyloid accumulation, and to assess the effects of transthyretin stabilization therapy on circulating neutrophil activation and inflammasome activity.
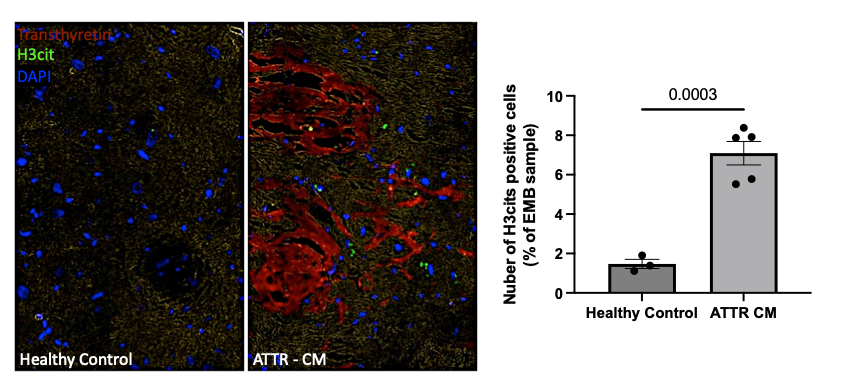
Methods Endomyocardial biopsies from age-matched ATTR-CM patients and healthy controls were analyzed using immunofluorescence microscopy for DNA (DAPI, blue), citrullinated histone H3 (H3Cit, green), and myeloperoxidase (MPO, red). NET deposition was quantified as the percentage of H3Cit-positive area relative to total area per field of view. Spatial analyses were performed to determine the proximity of activated neutrophils to transthyretin (TTR) deposits. In parallel, peripheral blood neutrophils were isolated from ATTR-CM patients before and after initiation of TTR stabilizer therapy, and from healthy controls. NET formation (spontaneous and PMA-stimulated) was assessed by immunofluorescence, and inflammasome activation was evaluated via ASC speck formation.
Results ATTR-CM samples showed markedly increased neutrophil infiltration (7.1 ± 1.3 % vs. 1.47 ± 0.4 %; p = 0.003) and NET deposition (315.8 ± 292.9 vs. 51.3 ± 34.5 µm²/FOV; p = 0.0097) compared with controls. Spatial analysis revealed that activated neutrophils were significantly closer to TTR deposits (16.1 ± 95.8 µm vs. 241.6 ± 368.5 µm; p < 0.0001), suggesting direct interaction between neutrophils and amyloid fibrils.Ex vivo analyses demonstrated increased spontaneous NETosis (5.7 ± 4.6 % vs. 0.74 ± 0.19 %; p = 0.007) and enhanced NET formation upon stimulation (45.1 ± 14.8 % vs. 17.2 ± 12.4 %; p < 0.001) in ATTR-CM patients compared with healthy controls. Neutrophils also displayed elevated ASC speck formation, indicating augmented inflammasome activation. After six months of TTR stabilizer therapy, stimulated NETosis was significantly reduced (44.7 ± 18.4 % vs. 8.0 ± 8.2 %; p = 0.006).
Conclusion Our findings reveal that neutrophil activation and NET deposition are prominent features of ATTR-CM and spatially associated with amyloid accumulation, implicating NET-driven inflammation in disease progression. Transthyretin stabilization therapy attenuates neutrophil activation and inflammasome activity, highlighting a potential therapeutic avenue targeting innate immune mechanisms to mitigate adverse cardiac remodeling in ATTR-CM.