Background
Asprosin is a recently discovered adipokine released by white adipose tissue during fasting. It is generated together with fibrillin-1, an extracellular matrix glycoprotein, through furin-mediated C-terminal cleavage of profibrillin encoded by the FBN1 gene. Elevated circulating asprosin levels have been linked to metabolic disorders and pro-inflammatory effects. Asprosin has been found in cardiac tissue (1). Here, we investigate which of the so far known asprosin receptors is expressed in healthy human cardiac tissue and whether their expression is age-dependent.
Methods
Asprosin localization was examined in human heart tissue (healthy donors and cardiac surgery patients), as well as in iPSC-derived cardiomyocytes. Immunostaining was performed using a custom-generated (Lab-Aspr) and the Birdy-1 (Novus Biologicals) anti-asprosin antibody. Expression of FBN1 and the asprosin receptors OR4M1, PTPRD, and TLR4 in human heart tissue from young (29 years) and older (59 years) donors (n = 12) was assessed by qPCR, normalized to the housekeeping genes IPO8 and GAPDH.
Results
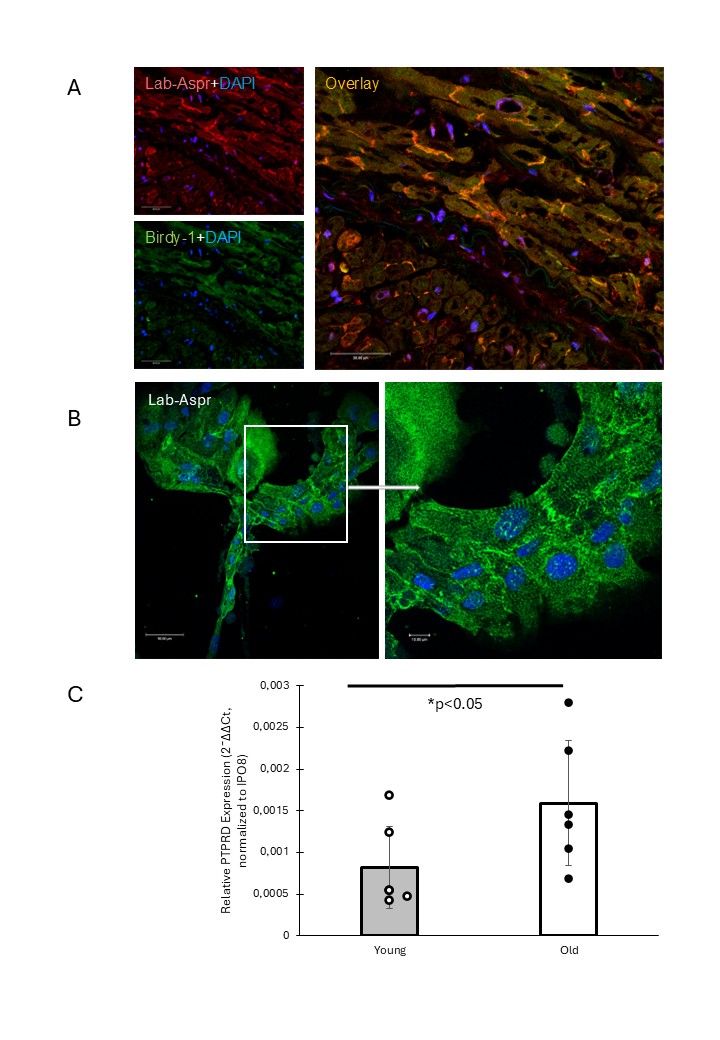
We confirmed the presence of asprosin protein in human cardiac tissue using two independent anti-asprosin antibodies. The signals from both antibodies overlapped and showed strong immunoreactivity localized to intercalated discs (Fig. 1A). Immunohistochemistry on human iPSCs showed diffuse asprosin distribution in undifferentiated cells. However, when these cells were differentiated into cardiomyocytes, the asprosin signal appeared in a striated pattern. Asprosin was predominantly found in intercalated discs (Fig. 1B). The expression of the asprosin precursor profibrillin was confirmed by qPCR. Furthermore, using immunohistochemistry and qPCR, we demonstrated that all so far known asprosin receptors—OR4M1, PTPRD, and TLR4—are expressed in human cardiac tissue. Of note, the asprosin receptor PTPRD was upregulated with aging. (Fig. 1C).
Conclusions
This study provides evidence that human heart possesses an intrinsic asprosin signaling system, including asprosin synthesis by cardiomyocytes, structural accumulation in cardiomyocytes and endothelial cells, and expression of all three asprosin receptors. The recruitment of asprosin into sarcomeric and junctional domains suggests involvement in cardiomyocyte maturation and intercellular communication. Furthermore, the observed age-dependent upregulation of PTPRD indicates that asprosin signaling may be dynamically regulated during aging, potentially linking metabolic and inflammatory pathways to age-related cardiac remodeling. In future experiments we will analyze the formation, release, and effect of asprosin on cardiomyocytes during mechanical work.
References
1 Beck C, Morcos Y A T, Piekarek N, Juette T, Gerken A, Lubomirov L T, Schroeter M M, Winkelmann A, Grisk O, Albes J M, Sengle G, Ladilov Y, Pryymachuk G. Asprosin as a potential new paracrine regulator in the heart. Cardiovasc Res 2024;120(Suppl 1):cvae088.217. doi:10.1093/cvr/cvae088.217.