Background: Clinical studies indicate that heart failure (HF) patients have a higher risk of developing cancer than patients without HF, especially lung cancer. On the other hand, cancer cachexia can contribute to cardiac remodelling. Mechanistically, this bidirectional interaction between HF and cancer is not yet fully understood.
Aims: This study aims to investigate the mechanisms of how HF promotes tumor progression, and how tumor can exacerbate HF.
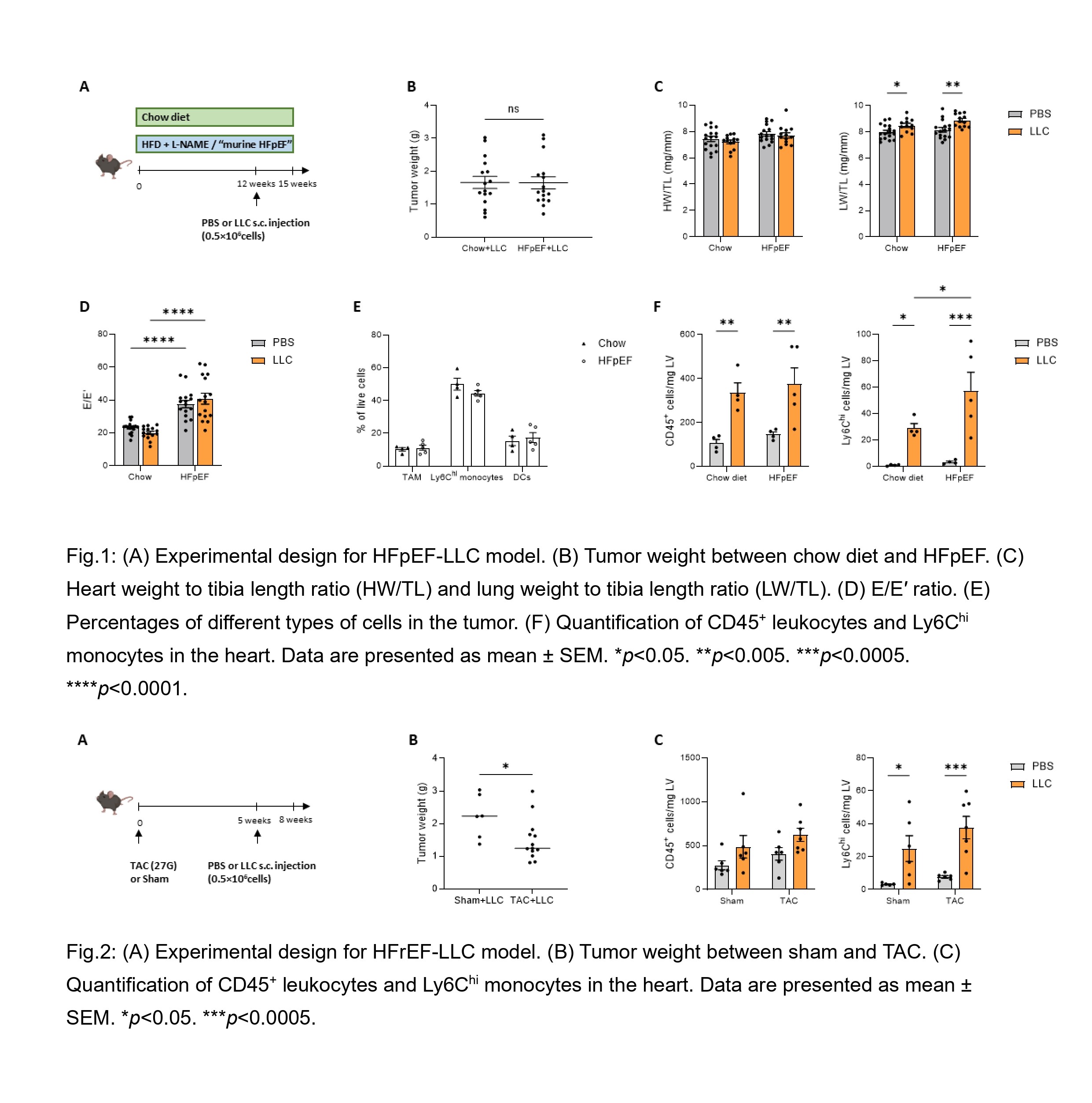
Methods: Male C57BL/6N mice were used to induce HF with preserved ejection fraction (HFpEF) by feeding high-fat diet and 0.5g/L L-NAME for 15 weeks. For HF with reduced EF (HFrEF) transaortic constriction (TAC) was performed. 0.5×10
6 Lewis Lung Cancer (LLC) cells suspended in 100μL of PBS were injected subcutaneously into the right flank vs. PBS alone. Mice were sacrificed 3 weeks after LLC (or PBS) injection (Fig. 1A and 2A). Cardiac function was evaluated using echocardiography. Immune cell infiltration in the heart and tumor was analysed by flow cytometry.
Results: Tumor weight did not differ between chow diet mice and HFpEF mice (Fig. 1B). There was no difference in cardiac hypertrophy (heart weight to tibia length ratio) among the groups. However, the lung weight to tibia length ratio significantly increased in tumor-bearing mice compared to non-tumor mice, independent of HF status (Fig. 1C). HFpEF mice had impaired diastolic function, as indicated by an elevated E/E′ ratio which was not altered by LLC (Fig. 1D). Flow cytometry revealed no significant differences in tumor-associated macrophages (TAM), Ly6C
hi monocytes, and dendritic cells (DCs) in the tumor microenvironment (Fig. 1E), but CD45
+ leukocytes and Ly6C
hi monocytes markedly increased in the hearts of tumor-bearing mice compared to non-tumor mice (Fig. 1F). Notably, HFpEF mice with tumor had a further significant increase in Ly6C
hi monocytes in the heart than chow diet mice with tumor. Surprisingly, in murine HFrEF induced by TAC, mice showed lower tumor weight compared with sham-operated mice (Fig. 2B). In the hearts of tumor-bearing mice, Ly6C
hi monocytes were also increased compared to non–tumor mice, whereas CD45⁺ leukocytes levels remained unchanged (Fig. 2C). Together, these results suggest a distinct inflammatory phenotype of the failing heart induced by concomitant tumor disease dependent on the different types of HF.
Conclusions: HFpEF does not accelerate solid tumor growth, however, HFrEF reduces tumor progression in mice. Tumor disease enhances cardiac immune cell infiltration in two separate models of HF.