Background Persistent immune activation contributes to left ventricular (LV) dysfunction and adverse remodeling in hypertrophic obstructive cardiomyopathy (HCM). Neutrophils drive chronic low-grade inflammation by releasing neutrophil extracellular traps (NETs), which can exacerbate tissue injury and inflammation.
Aim To investigate the chemotactic accuracy and NETotic activity of peripheral blood neutrophils in patients with hypertrophic obstructive cardiomyopathy (HOCM) and to examine their relationship to disease progression.
Material & Methods Peripheral blood neutrophils were isolated using the MACSxpress® Neutrophil Isolation Kit (Miltenyi Biotec) from patients with hypertrophic cardiomyopathy (HCM) with and without left ventricular outflow tract (LVOT) obstruction. Chemotactic behavior was assessed using the ibidi μ-Slide Chemotaxis assay with N-formyl-methionyl-leucyl-phenylalanine (fMLP; 100 nM and 300 nM) as chemoattractant. Live-cell imaging over 4 hours was used to quantify individual cell trajectories, including mean velocity, track straightness, cumulative cell area, and directional accuracy (rose plot analysis). NETosis assays were performed ex vivo, and neutrophil morphology was quantified by automated microscopy, classifying lobulated, delobulated, diffuse NETs, and spread NETs.
Results Neutrophils from HOCM patients had significantly reduced directional chemotactic accuracy for both chemoattractant doses. Cumulative cell area followed a sigmoid rather than exponential pattern, consistent with defective chemotactic efficiency. Ex vivo, neutrophils were primed for spontaneous NETosis. Shear stress–induced inflammation was observed, with the level of NETosis correlating with the increasing left ventricular outflow tract (LVOT) gradient in the affected patient.
Conclusions HOCM is associated with reduced neutrophil chemotaxis and priming for NETosis. Our results suggest, that neutrophils drive low-grade inflammation in HOCM.
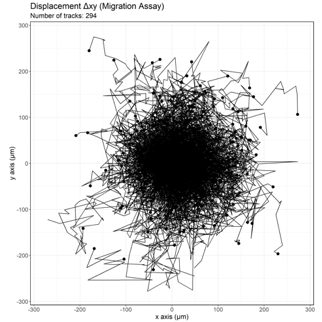
Figure: Rose plot showing cell tracks of neutrophils in HOCM patient under fMLP (300nM) stimulus with a random migration pattern and minimal directional displacement
