Background
Data on fatal complications following catheter ablation for atrial fibrillation (AF) remain limited, particularly with respect to different energy strategies and catheter designs. Microelectrode radiofrequency (RF) catheters enable very high-power, short-duration ablation (vHPSD, 90W, 4s), but the safety profile in real-world settings has not been fully characterised.
Objective
The aim of the TiFFANY (Total number of Fatal adverse events Following Atrial fibrillation ablatioN from an FDA mandatory reporting sYstem) micro study was to evaluate evaluate reported fatal events associated with microelectrode RF catheter ablation for AF, based on cases documented in the U.S. FDA Manufacturer and User Facility Device Experience (MAUDE) database.
Methods
The MAUDE database was queried for reports of deaths related to AF ablation procedures performed with RF energy between January 2014 and October 2025. All reports were manually reviewed. Duplicate records were removed. Cases involving a microelectrode RF ablation catheter were included in this analysis. When available, supplementary clinical details were extracted from referenced source materials. Event characteristics, diagnostic evaluation, and management strategies were presented descriptively.
Results
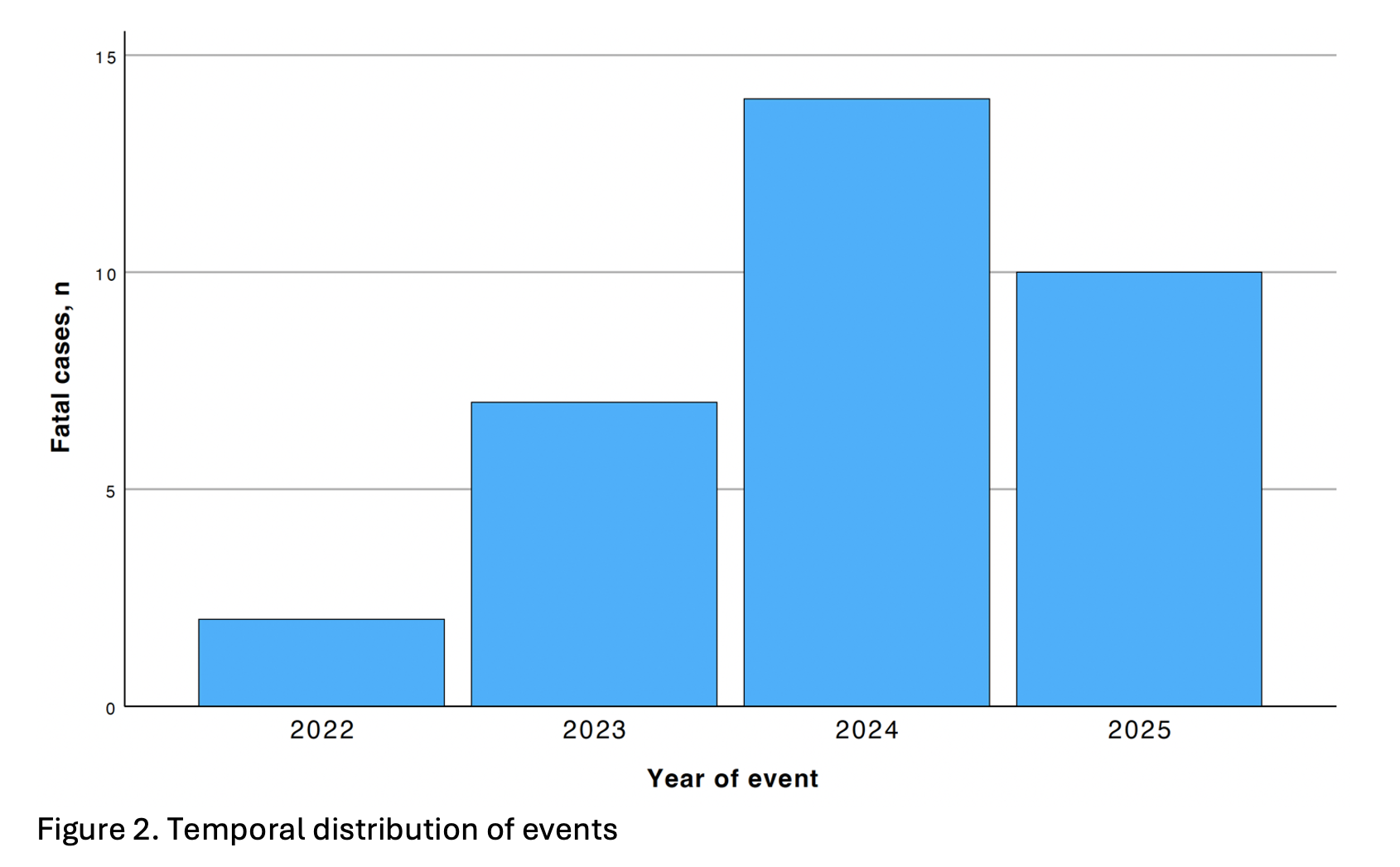
A total of 571 death reports related to catheter ablation for AF were identified in the MAUDE database. Among these, 174 unique cases involved RF ablation, of which 37 were associated with the use of a microelectrode RF catheter. The first fatal event linked to this technology was reported in December 2022; since then, microelectrode RF catheters have accounted for 48.7% (37/76) of all RF-related death reports.
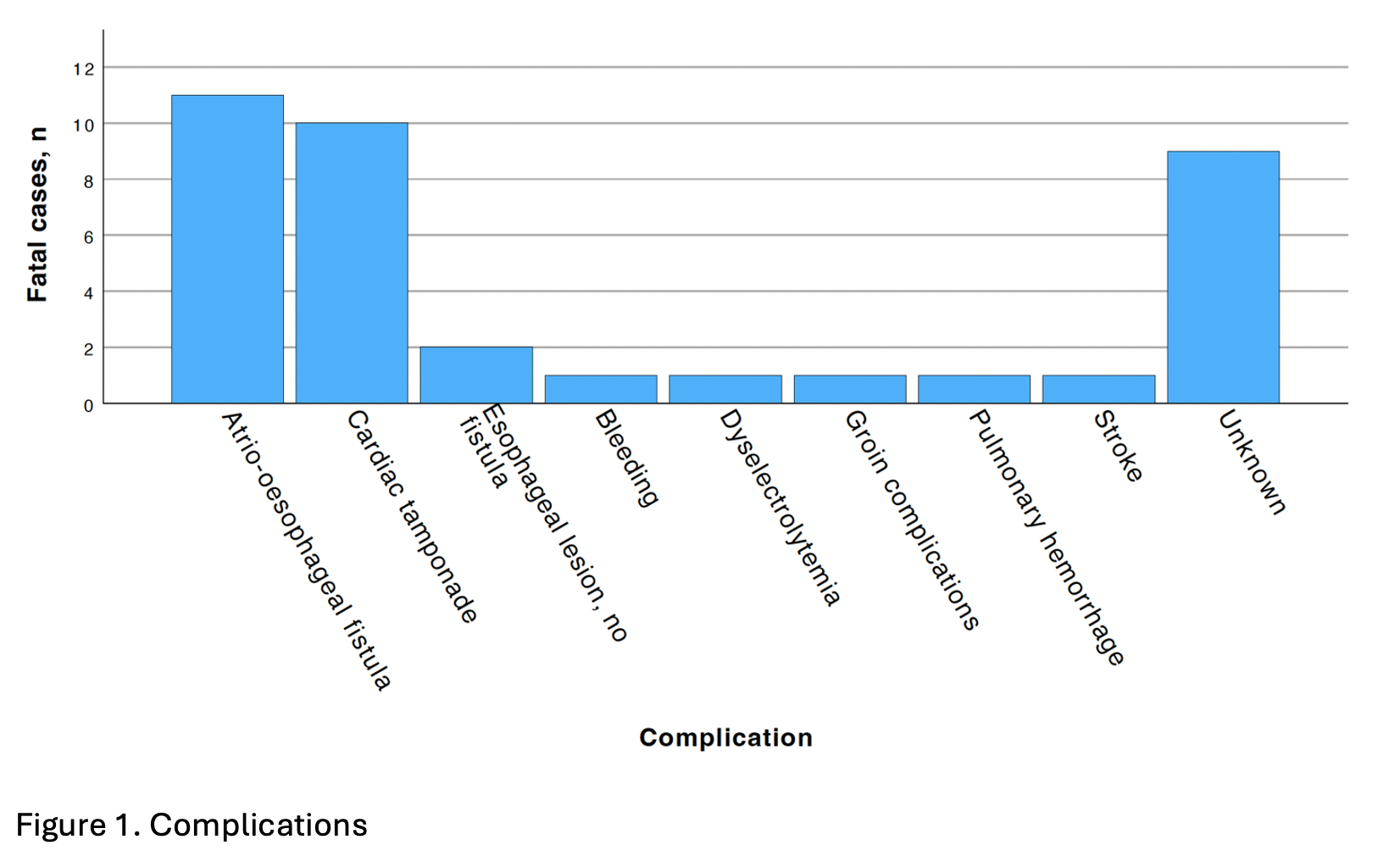
Information on the fatal complication was available in 75.7% (28/37) of cases. Atrio-oesophageal fistula was the most frequently reported cause of death (39.3%; 11/28), followed by cardiac tamponade (35.7%; 10/28). Most cases originated from the United States (57.1%; 20/35), followed by Japan (11.4%; 4/35). All events were documented as adverse clinical outcomes; none reported a confirmed device malfunction, and no catheters were available for manufacturer evaluation.
Symptom presentation was reported in 40.5% (15/37) of cases, most commonly hypotension (66.0%; 9/15). Diagnostic evaluation was documented in 51.4% (19/37) of cases, with echocardiography used in 78.9% (15/19) and computed tomography in 26.3% (5/19). No case reported endoscopic assessment. Treatment information was available in 51.4% (19/37) of patients; of these, 57.9% (11/19) underwent interventional management and 26.3% (5/19) surgical intervention.
Conclusions
Fatal events associated with microelectrode RF AF ablation appear disproportionately represented in recent MAUDE reports. These results must be interpreted with caution due to inherent MAUDE reporting bias, particularly variable manufacturer reporting and incomplete case information. Atrio-oesophageal fistula and tamponade were the leading death causes. No device malfunctions were identified. The data highlight the need for strengthened surveillance and improved safety strategies for vHPSD ablation.