Background:
Heart failure following myocardial infarction (MI) remains a major clinical challenge despite current therapies. The mitochondrial pyruvate carrier (MPC1) links glycolysis and oxidative phosphorylation by transporting pyruvate into mitochondria. Reduced MPC1 expression lowers mitochondrial oxygen consumption and increases pentose phosphate pathway activity, promoting anabolic metabolism. Previous studies have shown that MPC1 knockdown in cardiomyocytes can induce cardiac growth and hypertrophy, suggesting an adaptive metabolic response under stress.
Aim:
To investigate whether reduction of MPC1 protein expression prior to MI impacts myocardial injury and cardiac function.
Methods:
A tamoxifen-inducible, cardiomyocyte-specific Mpc1 knockout mouse model was used. Animals were heterozygous for Cre and Mpc1 (Cre het/Mpc1 het), and compared to littermates heterozygous for Cre but wild type for Mpc1 (Cre het/Mpc1 wt) as controls. Mice were between 8 and 13 weeks of age. Tamoxifen (20 mg/kg, i.p.) was administered for 5 consecutive days (days −5 to 0) to induce Mpc1 depletion before permanent ligation of the left anterior descending coronary artery (LAD). Echocardiography was performed at baseline and 5 days post-MI to assess left ventricular ejection fraction. At day 5, hearts were collected for histological and biochemical analyses. Mitochondrial respiration was evaluated using high-resolution Oroboros respirometry.
Results:
Preliminary echocardiographic data showed a reduction in ejection fraction in both MI groups compared to their respective sham groups. Analysis of mitochondrial function revealed reduced respiration in the Mpc wt group following MI intervention in comparison to the respective sham group. In contrast, respiration in the sham Mpc1 het group was already significantly reduced compared to the sham Mpc1 wt group and did not further decline upon MI induction (Figure 1). The study is still ongoing and data are being collected and analysed to substantiate these initial observations.
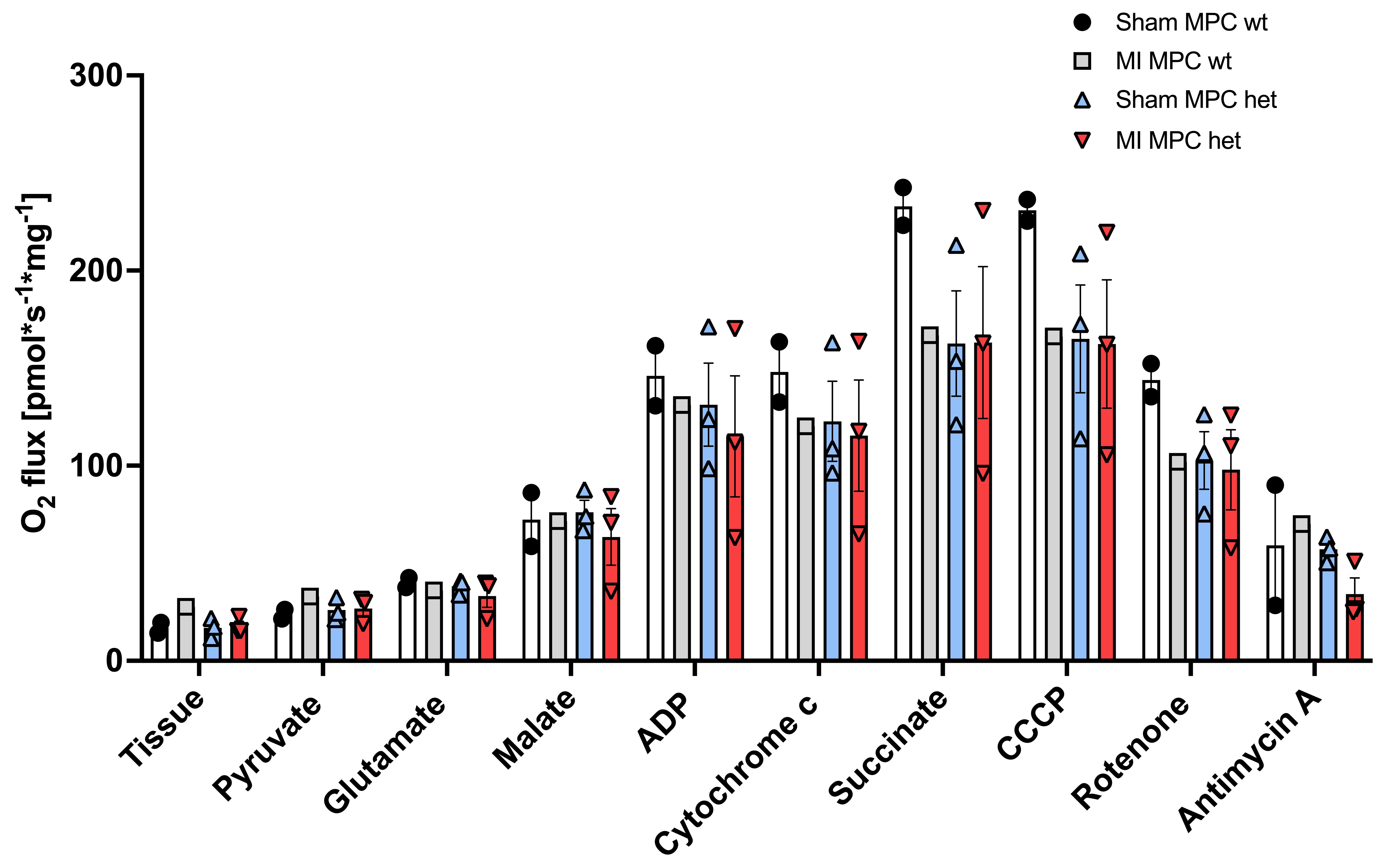
Figure 1: High-resolution respirometry assessment of mitochondrial activity.
