Aims: Atrial cardiomyopathy (AtCM) is linked to incident atrial fibrillation (AF), progression from paroxysmal to persistent AF, and recurrence after pulmonary vein isolation (PVI). Electroanatomical mapping (EAM) and cardiac MRI characterize AtCM but are resource-intensive. We evaluated non-invasive markers from ECG, transthoracic echocardiography (TTE), and blood biomarkers for (i) association with left-atrial low-voltage areas (LVA) on EAM and (ii) prediction of post-PVI arrhythmia recurrence.
Methods: We analyzed 200 consecutive first-time PVI patients in sinus rhythm. ECG metrics: P-wave duration (PWD), amplified PWD (APWD), P-wave dispersion, axis, P-terminal force in V1 (PTFV1), and P-wave amplitude in lead I. TTE measured left-atrial volume index (LAVI), left-atrial diameter (LAD), left-ventricular ejection fraction (LV-EF), and valve regurgitation. hs-troponin T (hs-TnT) and hs-CRP were obtained. LVA were defined as bipolar voltage <0.5 mV and staged by affected surface: I <5%, II ≥5–<20%, III ≥20–<30%, IV ≥30%. Univariable and multivariable logistic regression identified markers of advanced LVA (Stages III–IV, ≥20%). AF recurrence within 3–12 months was captured via structured phone follow-up and ECG documentation; the earliest evidence in this window defined recurrence (binary endpoint). Median follow-up was 277 days (IQR 210–360).
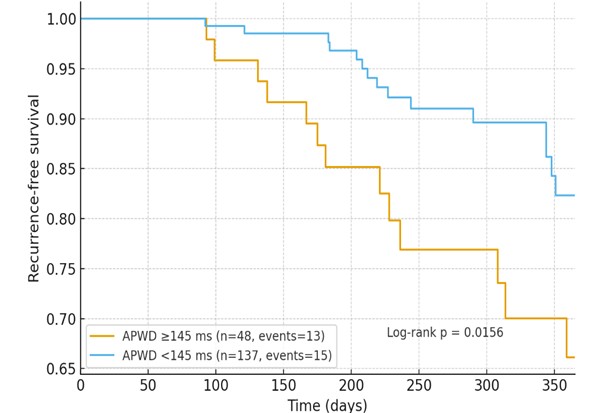
Results: Stages: I n=93, II n=69, III n=15, IV n=23. Advanced stages were associated with older age (Stage I 64 [60–71] vs. Stage IV 72 [68–80] years) and female sex (15.1% vs. 69.6%; both p<0.001). APWD rose stepwise (130 [120–137] → 163 [145–177] ms; p<0.001), while P-wave amplitude in lead I fell (0.09 [0.06–0.10] → 0.06 [0.04–0.08] mV; p<0.001). LAVI increased (37 [30–45] → 48 [42–60] mL/m²; p=0.002) and LV-EF was lower (62 [57–62]% → 57 [46–62]%; p=0.043). Univariable (per 1 SD for continuous; per 1 for binary): age OR 2.34 (95% CI 1.57–3.51), female sex 4.49 (2.14–9.40), CHA₂DS₂-VA 2.26 (1.50–3.41), hs-TnT 1.52 (1.10–2.10), APWD 3.01 (2.00–4.52), lower P-wave amplitude lead I 0.39 (0.22–0.67), pathologic P-axis 2.46 (1.08–5.64), LAD 1.51 (1.01–2.26), LAVI 1.99 (1.27–3.11), LV-EF 0.69 (0.50–0.97); relevant (≥moderate) mitral regurgitation was also associated. In multivariable analysis, female sex 22.55 (3.57–142.63; p<0.001), APWD 2.66 (1.23–5.75; p=0.013), and P-wave amplitude lead I 0.20 (0.05–0.81; p=0.024) remained independently associated. Kaplan–Meier (≤365 days) showed lower recurrence-free survival for APWD ≥145 ms vs. <145 ms (log-rank p=0.0156).
Conclusion: Among non-invasive AtCM markers, APWD and P-wave amplitude in lead I independently identified advanced AtCM; of these, only APWD also predicted post-PVI AF recurrence. ECG-based assessment—particularly APWD—enables early, non-invasive risk stratification and may reduce the need for invasive electroanatomical mapping.