Background. Inclisiran, a small interfering RNA targeting PCSK9, has demonstrated durable LDL-C lowering in clinical trials. However, real-world evidence on its long-term effectiveness and adherence remains limited.
Aim. To evaluate LDL-C reductions, adherence, and reasons for therapy discontinuation over a follow-up period of up to 3 years in a real-world lipid clinic cohort.
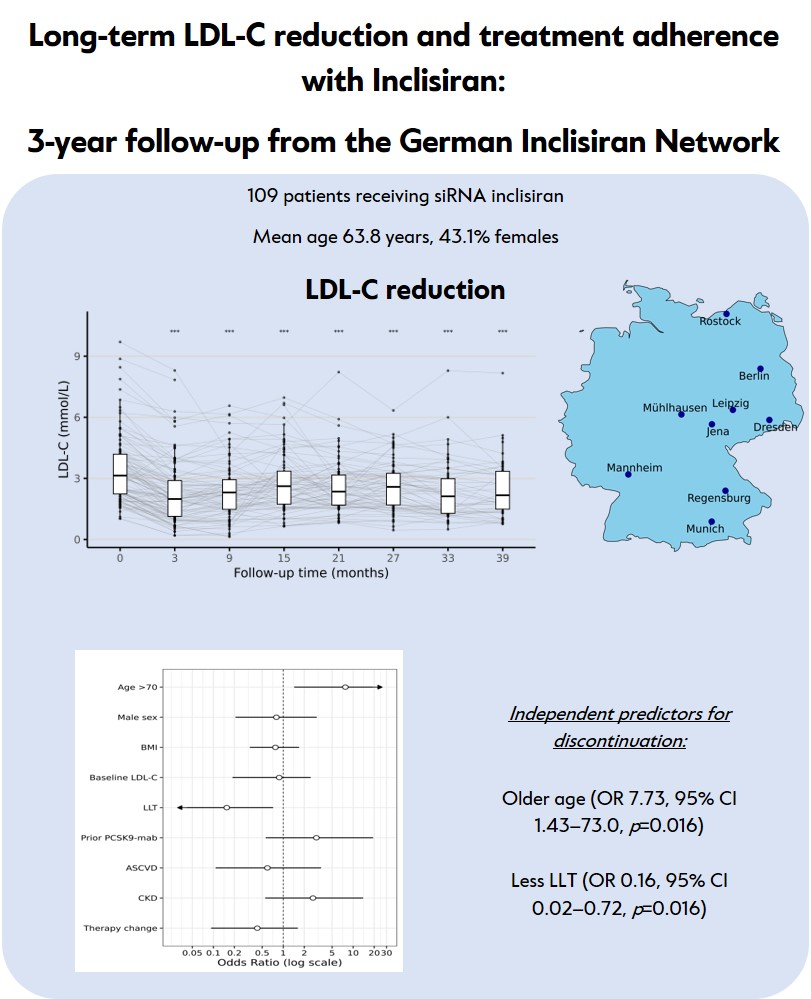
Methods. We analyzed data from ten lipid clinics participating in the German Inclisiran Network (GIN). Adults with at least one inclisiran dose and follow-up lipid measurements were included. LDL-C levels were assessed during three years of follow-up (median 33 months, IQR 27-39). Discontinuation rates, therapy changes, and adverse effects were documented.
Results. Among 109 patients (mean age 63.8 years, 43.1% female). The cohort comprised was heterogenous population, with only ~30% receiving statins due to statin intolerance. Median (95% CI) LDL-C reduction from baseline to nadir was -46.0% (-51.6% to -41.6%). Median for LDL-C reduction for each individual during the follow-up was -28% (-33.2% to-24.2%). High interindividual and intraindividual variability in LDL-C response was observed. Over three years, 28.4% of patients discontinued inclisiran, most commonly due to lack of efficacy, adverse effects or combination of both. In a multivariable logistic regression model, older age was a predictor of higher discontinuation rates, whereas concomitant LLT use was associated with lower discontinuation rates.
Conclusions. In a heterogeneous real-world population, inclisiran achieved significant LDL-C reductions but with marked inter- and intraindividual variability. A notable proportion of patients discontinued therapy, highlighting the importance of individualized treatment strategies.