Background
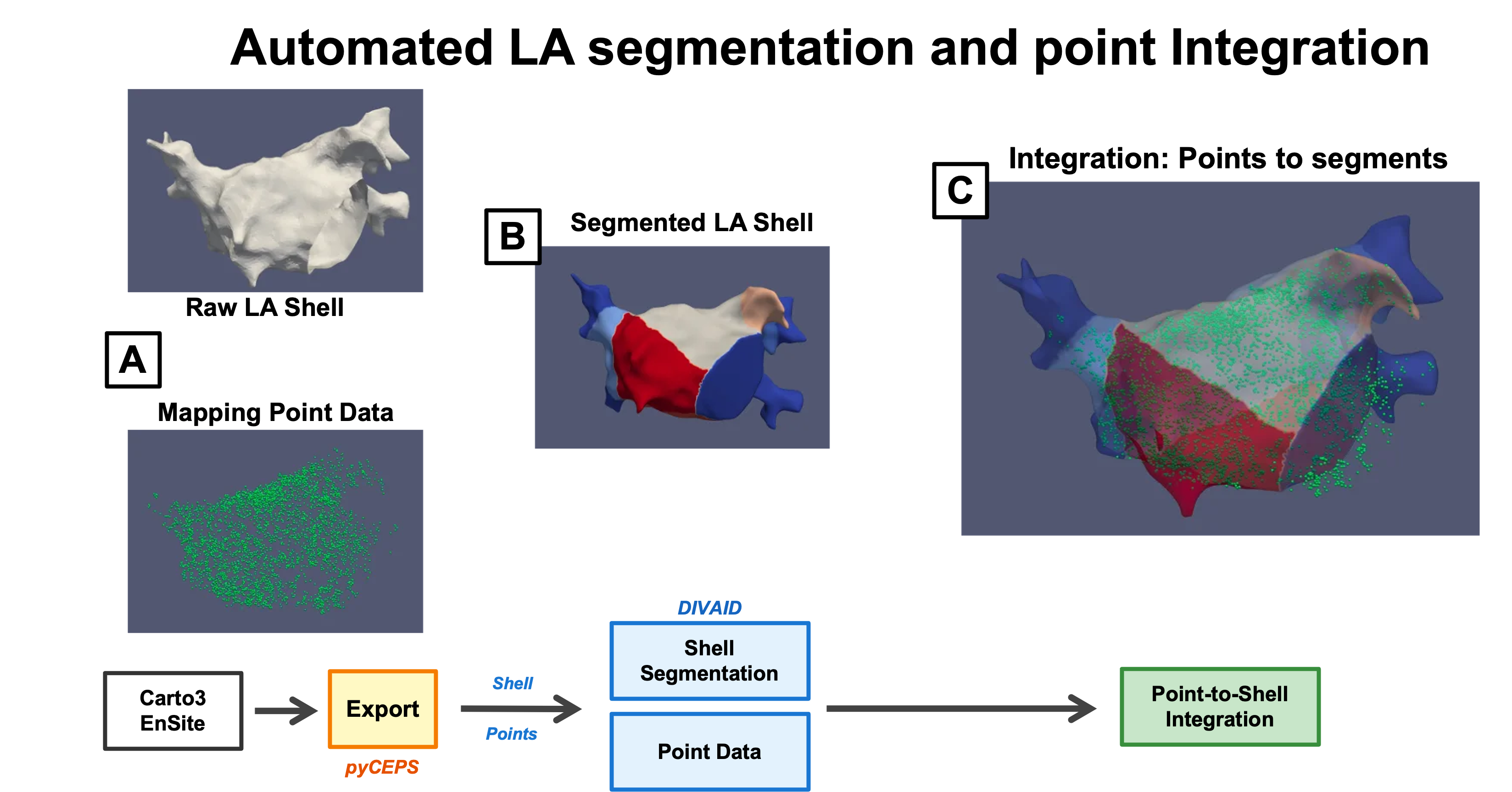
Diffuse interstitial fibrosis is a key arrhythmogenic substrate for atrial arrhythmias (AAs) but may be under-detected by commonly used bipolar voltage thresholds in electroanatomical mapping (EAM). In LV cardiomyopathy (LV-CM) averaged unipolar voltage inversely reflects viable myocardium and relates to interstitial fibrosis and heart‑failure prognosis in VT cohorts, enabling genotype‑aware substrate assessment beyond binary bipolar cutoffs in LV-CM. We present, to our knowledge, the first clinical application of the EHRA 2025 left‑atrial (LA) regionalization to EAM, implementing an automated postprocessing workflow that segments the LA and aggregates mapping points to regions to derive segmental unipolar voltage metrics.
Purpose
To develop and evaluate an automated workflow for LA segmentation and to compare regional unipolar voltage patterns across LV-CM genotypes.
Methods
EAM data of LV-CM patients undergoing CA for atrial fibrillation (AF) or non CTI-dependent atrial tachycardia (AT) between 2016-2024 and four control patients without structural heart disease undergoing CA for paroxysmal AF (PAF) or focal AT were included. A postprocessing pipeline was developed to perform automated LA segmentation according to current consensus recommendations using the DIVAID tool. Individual mapping points were merged with segmented anatomical shells to enable region-specific voltage analysis.
Results Fifty-four LV-CM patients (age 57 ±13 years, 80% male, LV-EF 47% ±11%, LAVI 45±16ml/m2, GLS -14%±5%; Genetics: LMNA n=8; TTN n=6; gene elusive and other LP/PV genotypes n=40) were included yielding 69,389 mapping points. Four control patients (age 46±13 years, normal LV-EF, LAVI 27±3ml/m2) provided 16,768 points. Automated segmentation succeeded in all cases. Median unipolar voltage was lower in LV-CM than controls (1.55 mV, IQR 0.8–2.88 vs 3.11 mV, IQR 1.94–4.87; p<0.001) and was reduced across all anatomical segments. In genotype-specific subanalysis, LMNA showed the lowest voltages (1.48 mV, IQR 0.86–2.56), whereas TTN was more preserved (2.17 mV, IQR 1.13–3.21); LMNA < TTN overall (p<0.001). Segment-wise, LMNA was lower in all regions except the posterior wall and left veno-atrial junction.
Conclusion Automated EHRA-based LA regionalization enables systematic regional unipolar analysis. LV-CM exhibits globally reduced atrial unipolar voltages, with pronounced genotype-specific differences. This approach supports unipolar mapping as a substrate biomarker beyond bipolar voltage thresholds and may provide novel methodology for prognostic assessment in AF care.