Background and aim: Chronic, low-grade inflammation contributes significantly to the development and progression of heart failure (HF). We tested the hypothesis that there is a reciprocal interaction between neutrophils (N) undergoing NETosis and the activation of macrophages (Mf) in the myocardium of patients with HF.
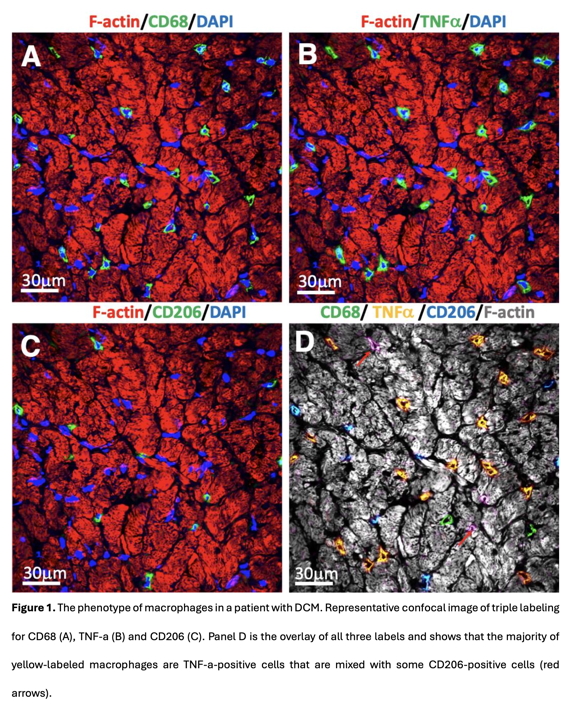
Methods: Ventricular biopsies from patients with HF due to dilated (DCM, n=7), ischemic (ICM, n=7), and inflammatory (InfCM, n=7) cardiomyopathy, and 5 control patients were studied. N undergoing NETosis were quantified using a triple labeling with CD66b, citrullinated histone 3 (citH3), and myeloperoxidase (MPO). The total number of Mf was determined using a CD68 antibody. M1 macrophages were determined by triple labeling with CD68 and TNFalpha, IL-1beta, and IL-6. M2 macrophages were determined using CD68, arginase I, and CD206 (Figure 1).
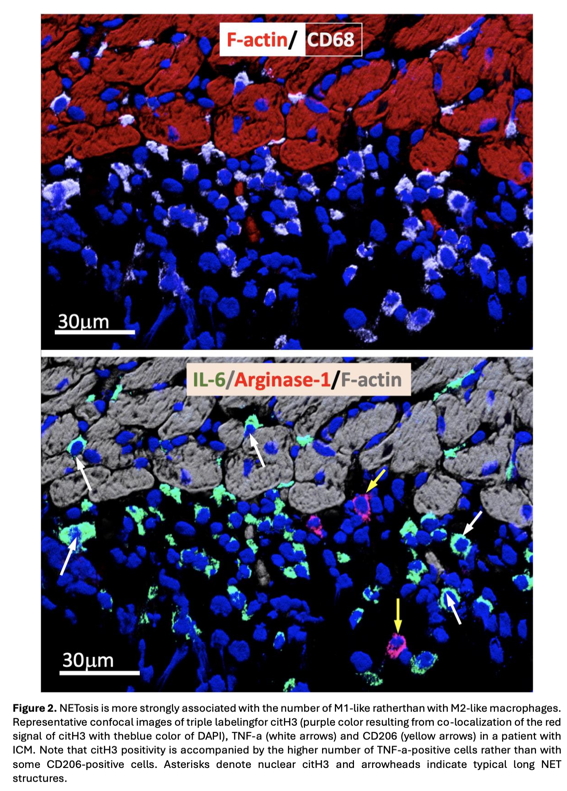
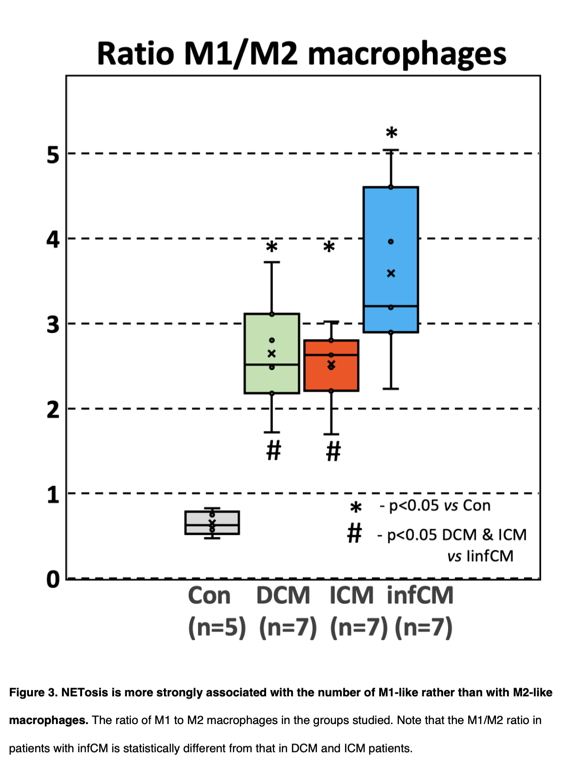
Results: The number of N per myocardial area in HF groups was 3- to 4-fold higher than in control hearts. Quantification of N undergoing NETosis revealed a median of 49% [IQR, 45-52] in infCM, 45% [39-46] in ICM, and 41% [37-45] in DCM. In control patients, less than 4% [3.8-4.2] of N developed NETosis. One square mm of the myocardial surface in the control group comprised a median of 13 [7-18] Mf, whereas the same myocardial surface comprised a median of 49 [43-62] Mf in DCM, 57 [48-66] in ICM, and 58 [56-67] in infCM. The elevation in Mf count correlated very strongly (r=0.91, p < 0.01) with the number of N. Quantitative analysis of the percentage of M1 in infCM revealed a value of 78.5% [70–85] that differed significantly (p < 0.05) from the median percentage of M1 in patients with DCM (53% [48–5]4) or ICM (54% [51–60]). The median percentage of M1 Mf in the control group was 3% [1.6–5.5] and differed significantly (p < 0.01) from all HF groups. The median percentage of M2f was 21% [18–28] in DCM, 22% [18–22] in ICM, and 23% [21.2–25.1] in infCM, respectively. The median percentage M1 in the control group was 6.5% [4.8–7.6] and differed significantly (p<0.01) from all HF groups. Correlative analysis revealed a statistically significant positive correlation (r=+0.68, p<0.01) between M1 and the proportion of NETosis. In contrast, there was no significant correlation between M2 and the proportion of NETsis. Moreover, triple labeling of tissue sections with antibodies against citH3, TNF-alpha, and CD206 supported this observation and demonstrates that the citH3-positive cells are surrounded by many more TNF-alpha-positive Mf than CD206-positive Mf (Figure 2). The significantly higher increase in M1 compared to M2 Mf was reflected in the M1/M2 ratio (Figure 3). The median of this ratio was 3 [2.5–4.3] in patients with infCM and differed significantly (p < 0.05) from the median of the M1/M2 ratio in patients with DCM (2.51 [2–3]) and ICM (2.63 [2.3–2.8]). The median of the M1/M2 ratio in the control group was 0.63 [0.6–0.8] and differed significantly (p < 0.01) from all HF groups (Figure 3). In summary, these data indicate that, in the setting of HF, there is a shift in Mf activation towards a proinflammatory M1-like phenotype in connection with the increased NETosis.
Conclusion: Accumulations of N and Mf are typical features of chronic myocardial inflammation in HF, regardless of its etiology. An increased number of N undergoing NETosis in HF is associated with the activation of myocardial Mf and switches these cells towards a proinflammatory M1-like phenotype.