Background
Amyloid-transthyretin cardiomyopathy (ATTR-CM) is a systemic disease characterized by extracellular deposition of misfolded transthyretin fibrils, affecting myocardial structure and function as well as electric conduction. Due to a small ventricle and low stroke volume, ATTR-CM patients need a higher heart rate (HR) to maintain cardiac output. On the other hand, ATTR-CM patients often show autonomic neuropathy, bradycardia and conduction alterations, progressing with advancing disease. Our study aims to describe the association between Holter-ECG findings and biomarkers for risk stratification and management in ATTR-CM.
Methods
In this retrospective multi-centre cohort study, we included adult patients with confirmed ATTR-CM from the Comprehensive Heart Failure Center, University Hospital Würzburg, and the German Heart Centre Munich, who underwent Holter monitoring between 2017 and 2025.
We reviewed clinical data collected within six months before/after the Holter-ECG, including echocardiography, laboratory tests, and clinical evaluation. Holter reports, both in-house and external, were included. Descriptive statistics are presented as mean (SD) or median (quartiles). Associations between ECG, Holter, echocardiographic, and laboratory parameters were analyzed using SPSS. Significant correlations were further evaluated by linear regression.
Results
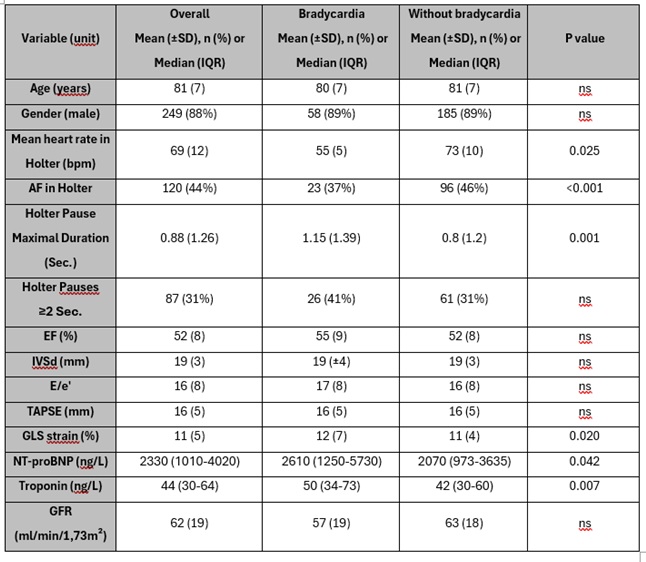
We included 282 ATTR-CM patients (249 (88%) male, 222 (79%) wild-type ATTR-CM). Mean left ventricular ejection fraction (LVEF) was 52 (8) %, interventricular septal thickness (IVSd) 19 (3) mm, and global longitudinal strain (GLS) −11 (5) %. Median NT-proBNP was 2330 ng/mL (1010- 4020), troponin T 44 ng/mL (30-64), and mean estimated glomerular filtration rate (eGFR) 57.3 (19) mL/min/1.73 m².
Holter monitoring revealed atrial fibrillation in 120 (44%) patients and pauses ≥ two seconds in 87 (31%) patients. A mean HR ≤ 60 bpm was observed in 57 (21%) patients, while only 37 (13%) had HR ≥ 80 bpm.
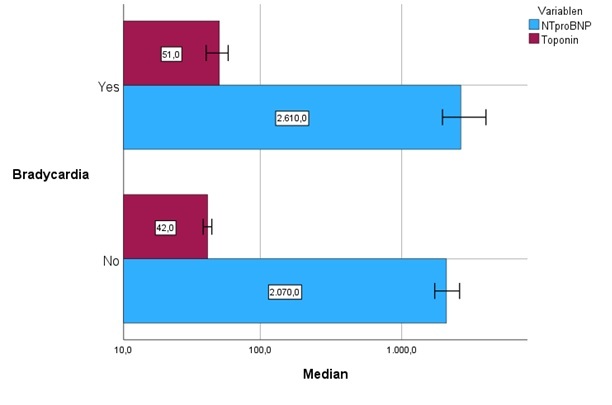
Bradycardia, defined as a mean Holter-ECG HR ≤ 60 bpm, was associated with higher levels of NT-proBNP and troponin T when compared to patients without bradycardia (table). Further, the presence of pauses ≥2 seconds were also associated with higher NT-proBNP levels (median 2700 ng/L (1905–4595) vs. 1710 ng/L (710–3773). Although a trend toward higher troponin T levels was observed in patients with pauses, it did not reach statistical significance (p = 0.074).
All significant associations remained robust after multivariable adjustment for age, amyloidosis subtype, eGFR, and LVEF. The adjusted models estimated a mean difference in NT-proBNP levels of 6000 ng/L in patients with vs. without pauses ≥2 seconds (p=0.002) and 7320 ng/L in those with bradycardia vs. non bradycardic patients (p=0.008). Bradycardic patients also had significantly higher troponin T (mean +10 ng/L; p=0.005).
Conclusion:
Holter monitoring in patients with ATTR-CM revealed a substantial share of patients with HR ≤ 60 bpm and with pauses of ≥2 seconds. Bradycardia and pauses strongly associate with biomarkers for risk stratification and management in ATTR-CM. Further, elevated biomarkers should trigger thorough rhythm monitoring to detect respective disorders.