Background
Recurrence after cryoballoon pulmonary vein isolation (PVI) remains common. How multiparametric procedural patterns interact with comorbidities and demographics to shape recurrence risk is unclear. We tested whether model-derived patient embeddings can uncover latent phenotypes linked to recurrence.
Objective
To identify clinically interpretable patient/procedural clusters associated with atrial fibrillation (AF) recurrence using unsupervised machine learning.
Methods
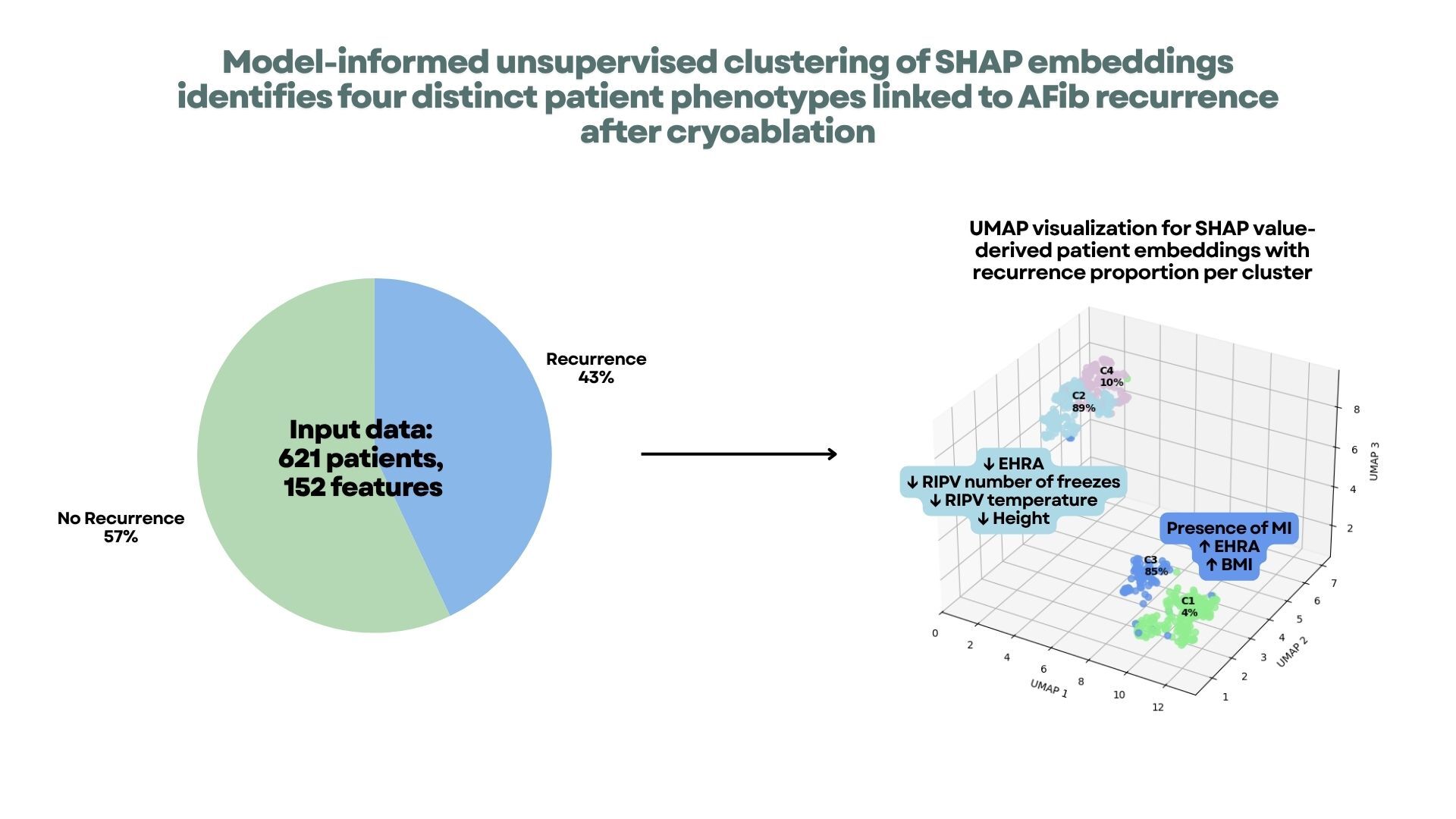
We analyzed 621 patients and 152 variables (demographics, comorbidities, medications, cryo parameters, outcomes). A supervised XGBoost model was trained to predict recurrence with the recurrence label used only as the target (not as a feature). Per-patient, per-feature Shapley values (SHAP) quantified feature contributions and served as embeddings for clustering. K-means on SHAP profiles identified clusters; the number of clusters was chosen using silhouette, Calinski–Harabasz, and Davies–Bouldin indices and corroborated with UMAP visualization. Cluster-level recurrence proportions and feature importance patterns were then summarized.
Results
AF recurrence occurred in 267/621 patients (43%),59.6% male; mean age 66.9 ± 11.0 years.
Four clusters were identified (Figure 1): C1 (n=197, 4% recurrence), C2 (n=190, 89%), C3 (n=89, 85%), and C4 (n=145, 10%).
-
C1 (low risk): longer total freeze time in RSPV/LIPV, digitalis use, and lower RSPV minimum freeze temperature; EHRA class and EP-lab duration were secondary.
-
C2 (high risk): lower symptom class (EHRA) and fewer RIPV freezes; mixed signals from ACE use, PV temperature metrics (LSPV/RSPV/RIPV), CRP, and height contributed to the inclusion in ths cluster.
-
C3 (high risk): prior myocardial infarction and higher BMI/weight; higher RIPV freeze temperature at 30 sec, fluoroscopy dose/time, longer procedure duration, and LIPV isolation temperature.
-
C4 (low–intermediate risk): prior cardioversion enriched; absence of cardioversion favored inlcusion in this cluster. Hemoglobin, weight, valve surgery, MCV, prior ablation, height, RSPV temperature, and creatinine were associated with the inclusiono in this cluster without a consistent direction.
Conclusion
Unsupervised learning on model-derived embeddings revealed four reproducible patient–procedure phenotypes with markedly different recurrence risks (4–89%). This approach captures multivariate, non-linear patterns—spanning cryo dosing, temperatures, and clinical comorbidity profiles—that are not readily detected with conventional univariate or standard multivariable analyses. If validated prospectively, such phenotyping could enable pre-procedural risk stratification, guide intraprocedural dosing strategies, and inform enrichment/stratification in future trials.