Synchronization and Control Framework for Panoramic Optical Mapping and Multi-View Motion Tracking of Mouse Hearts
Introduction
Panoramic optical mapping, which uses voltage-sensitive dyes to visualize transmembrane voltage on the beating heart, is inherently susceptible to motion artefacts which distort the fluorescence signal. While pharmacological uncouplers such as blebbistatin are used to minimize motion, they unfortunately interfere with cardiac electrophysiology, and hence the actual source signals. This limitation is compounded when mapping murine hearts due to their rapid cardiac cycle and small size, which pose additional technical challenges.
We developed a novel control framework that synchronizes multi-camera acquisition at high frame rates with a motion correction algorithm which substantially reduces motion-related noise and enables the accurate measurement of cardiac electrical signals from beating mouse hearts.
Methods
We used Langendorff-perfused, beating mouse hearts with voltage-sensitive dye Di-4-ANBDQPQ to visualize electrical excitation waves. To enable high-frame-rate synchronization, we developed a control framework with a Graphical User Interface (GUI) that automates the entire panoramic system, achieving microsecond-precision control over acquisition from 8 cameras, electrical stimulation, and light projection via 4 light-crafters through parallel processing on the microcontroller’s cores (cogs).
Following image acquisition, the image data were pre-processed, segmented, and pixel displacements were numerically tracked using optical flow-based algorithms with sub-pixel accuracy. The resulting motion-tracking data were then used to warp individual frames, which were subsequently compared with uncorrected data to evaluate motion-correction performance.
Results and Conclusions
Our control framework achieved precise synchronization of eight cameras operating at a maximum frame rate of 500 fps. The fluorescence signal exhibited intensity swings that were caused by motion of the cardiac substrate relative to the stationary camera pixels prior to correction (Figure 1A). Applying the motion correction algorithm successfully reduced motion artefacts to near-zero-pixel displacement, indicating effective motion removal (Figure 1B, compare average pixel displacement with motion, red, and motion-corrected, blue). When the fluorescence signal was replotted taking into account motion of the underlying substrate, it revealed sharply defined action potential signals post-correction (Figure 1C).
These results demonstrate that integrating the panoramic optical mapping system with an adaptive computer vision algorithm significantly reduces motion-related artefacts. This advancement enables accurate optical measurement of electrophysiological activity in beating mouse hearts, a key tool for basic and translational cardiac research, and provides the necessary stability required for advanced analyses tracking electrophysiology across the entire cardiac surface.
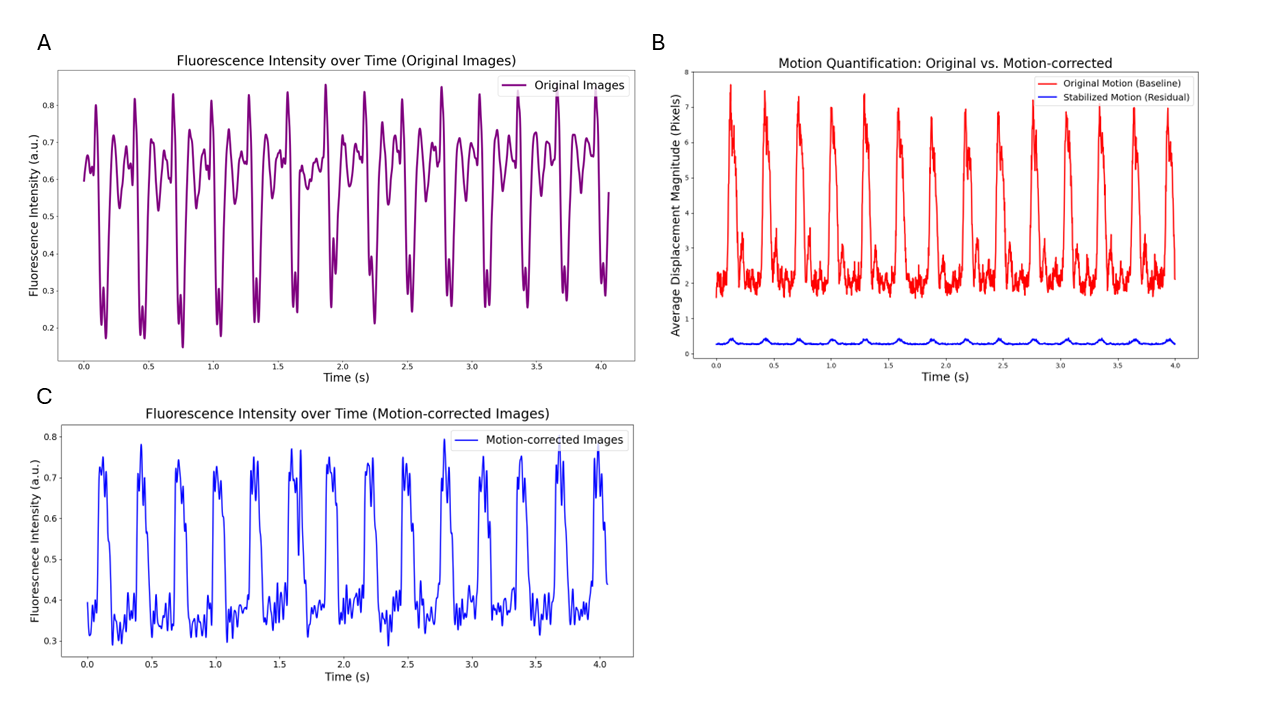
Figure 1 | (A) Original Fluorescence Intensity: Reflects electrically and mechanically induced intensity changes per pixel. (B) Motion Quantification: The average pixel displacement per frame was effectively reduced to sub-pixel levels (less than 1 pixel) by application of the motion correction algorithm. (C) Corrected Fluorescence Signal: Fluorescence Intensity plot after motion correction shows well-defined cardiac action potentials.
